Douglas Fearon
Professor
Cancer Center Member
M.D., Johns Hopkins University School of Medicine, 1968
dfearon@cshl.edu | 516-367-5420
Faculty ProfileI’m studying how to harness the power of the immune system to fight cancer. Our underlying premise is that the microenvironment within a tumor suppresses the immune system. We have found a way to eliminate this suppression in the mouse model of pancreatic cancer, which has led to development of a drug for human pancreatic cancer that will enter phase 1 clinical trials in 2015.
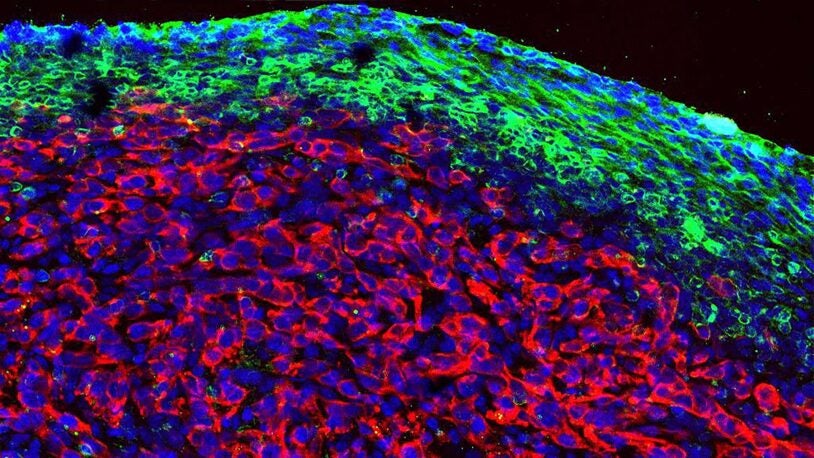
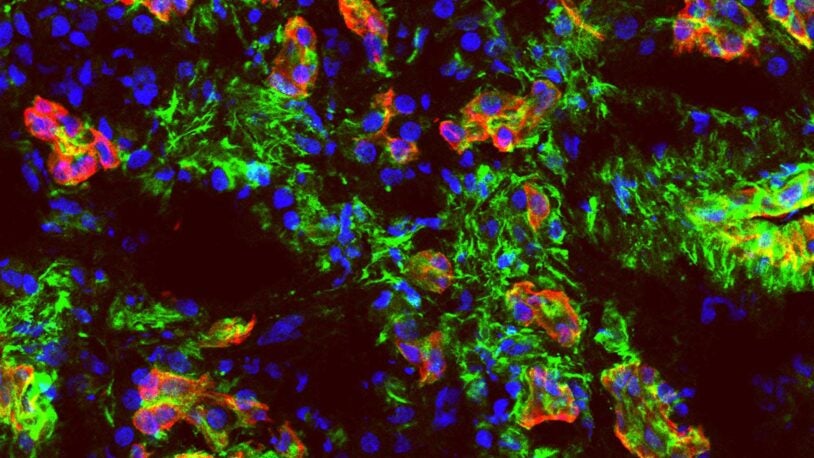
The Fearon laboratory studies the interaction between cancer and the immune system. Our underlying premise is that the tumor microenvironment is immune suppressive because cancer cells elicit responses characteristic of wound healing and tissue regeneration. This approach has led to the finding that activated fibroblasts in the tumor stroma mediate immune suppression in several mouse models of cancer, including the autochthonous model of pancreatic ductal adenocarcinoma of the Tuveson lab. Our understanding of the basis of immune suppression is evolving, but we know that it involves the production of the chemokine, CXCL12, by the fibroblastic stromal cells, binding of this CXCL12 by pancreatic cancer cells, and exclusion of T cells from the vicinity of the cancer cells. T cell exclusion, which also occurs in several types of human adenocarcinomas, causes antagonists of T cell checkpoints to be ineffective, despite the presence of cancer-specific CD8+ T cells. This immune suppression in murine and human pancreatic cancer has been interrupted by administering AMD3100, an inhibitor of the CXCL12 receptor, CXCR4, which leads to the rapid intra-tumoral accumulation of T cells. Since AMD3100 must be given by continuous intravenous administration, we are developing an anti-CXCR4 monoclonal antibody that would be administered every two weeks. Our next steps are to refine the necessary characteristics of this antibody.
Old chemo drug, new pancreatic cancer therapy?
August 26, 2024
CSHL scientists have found that folinic acid makes immunotherapy therapy more effective against pancreatic ductal adenocarcinoma in mice.
Can we crack this cancer’s immune response?
November 29, 2023
CSHL research brings scientists closer to understanding how pancreatic cancer interacts with our immune system and why immunotherapy hasn’t worked.
Cold Spring Harbor Laboratory 2022 Ph.D.’s
May 1, 2022
The School of Biological Sciences awarded Ph.D. degrees to ten students this year. Here are some stories and memories from their time at CSHL.
Deactivating pancreatic cancer’s protective coat of arms
February 17, 2022
Find out how cancer cells shield themselves from the immune system using a coat of armor and confusing signals.
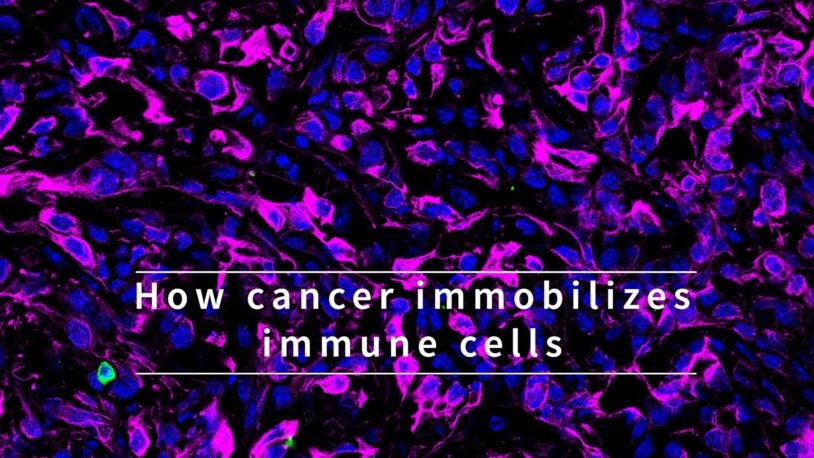
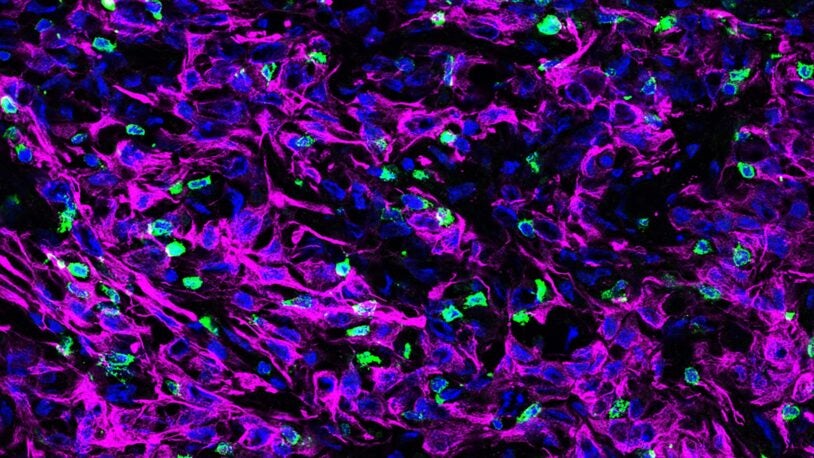
Pushing past pancreatic tumors’ defenses
February 16, 2022
A complex of proteins secreted by tumor cells can immobilize an important component of the immune system.
Giving the immune system a double boost against cancer
October 30, 2020
Immunotherapy enhances the body’s own defenses to kill cancer. A new regimen may increase the utility of these types of drugs.
Big bold dreamers
December 10, 2018
Dreamers drive discoveries. CSHL honors its many scientists making big impacts in their respective fields.
Base Pairs Episode 15.5: Cellular hide and seek
June 15, 2018
Learn how William Coley's daughter founded the Cancer Research Institute, Professor Fearon on the immune system, and white blood cells on screen.
How pancreatic cancer spreads after surgery
May 17, 2018
Researchers have solved a mystery about how pancreatic cancer spreads
How deadly cancer spreads after surgery
May 17, 2018
Scientists have solved an important mystery about how pancreatic cancer spreads to the liver even after a successful surgery.
Selected Publications
AMD3100/Plerixafor overcomes immune inhibition by the CXCL12-KRT19 coating on pancreatic and colorectal cancer cells.
26 Mar 2021 | British Journal of Cancer
Fearon, Douglas; Janowitz, Tobias;
CXCR4 inhibition in human pancreatic and colorectal cancers induces an integrated immune response
17 Nov 2020 | Proceedings of the National Academy of Sciences of the United States of America | 117(46):28960-28970
Biasci, D; Smoragiewicz, M; Connell, C; Wang, Z; Gao, Y; Thaventhiran, J; Basu, B; Magiera, L; Johnson, T; Bax, L; Gopinathan, A; Isherwood, C; Gallagher, F; Pawula, M; Hudecova, I; Gale, D; Rosenfeld, N; Barmpounakis, P; Popa, E; Brais, R; Godfrey, E; Mir, F; Richards, F; Fearon, D; Janowitz, T; Jodrell, D;
Cancer cell CCR2 orchestrates suppression of the adaptive immune response.
5 Oct 2020 | Journal of Experimental Medicine (JEM) | 217(10)
Fein, Miriam; He, Xue-Yan; Almeida, Ana; Bružas, Emilis; Pommier, Arnaud; Yan, Ran; Eberhardt, Anaïs; Fearon, Douglas; Van Aelst, Linda; Wilkinson, John; Dos Santos, Camila; Egeblad, Mikala;
CXCR4 inhibition in human pancreatic and colorectal cancers induces an integrated immune response
10 Jul 2020 | bioRxiv
Biasci, D; Smoragiewicz, M; Connell, C; Wang, Z; Gao, Y; Thaventhiran, J; Basu, B; Magiera, L; Johnson, I; Bax, L; Gopinathan, A; Isherwood, C; Gallagher, F; Pawula, M; Hudecova, I; Gale, D; Rosenfeld, N; Barmpounakis, P; Popa, E; Brais, R; Godfrey, E; Mir, F; Richards, F; Fearon, D; Janowitz, T; Jodrell, D;
Autophagy Promotes Immune Evasion of Pancreatic Cancer by Degrading MHC-I
May 2020 | Nature | 581(7806):100-105
Yamamoto, K; Venida, A; Yano, J; Biancur, D; Kakiuchi, M; Gupta, S; Sohn, A; Mukhopadhyay, S; Lin, E; Parker, S; Banh, R; Paulo, J; Wen, K; Debnath, J; Kim, G; Mancias, J; Fearon, D; Perera, R; Kimmelman, A;
All Publications
Identification of B cell antigens in solid cancer: initial insights and functional implications
28 Apr 2025 | Frontiers in Immunology | 16:1571570
Yang, Jung-In; Moresco, Philip; Fearon, Douglas; Yao, Min;
Signal peptide-independent secretion of keratin-19 by pancreatic cancer cells
20 Jan 2025 | bioRxiv
Moresco, Philip; Kastan, Jonathan; Yang, Jung-In; Prabakar, Rishvanth; Minicozzi, Francesca; Adams, Dexter; Cifani, Paolo; Tuveson, David; Fearon, Douglas;
Intratumoral NKT cell accumulation promotes antitumor immunity in pancreatic cancer
16 Jul 2024 | Proceedings of the National Academy of Sciences of the United States of America | 121(29):e2403917121
Li, Jiayun; Moresco, Philip; Fearon, Douglas;
Interaction between MED12 and ΔNp63 activates basal identity in pancreatic ductal adenocarcinoma
17 Jun 2024 | Nature Genetics
Maia-Silva, Diogo; Cunniff, Patrick; Schier, Allison; Skopelitis, Damianos; Trousdell, Marygrace; Moresco, Philip; Gao, Yuan; Kechejian, Vahag; He, Xue-Yan; Sahin, Yunus; Wan, Ledong; Alpsoy, Aktan; Liverpool, Jynelle; Krainer, Adrian; Egeblad, Mikala; Spector, David; Fearon, Douglas; Dos Santos, Camila; Taatjes, Dylan; Vakoc, Christopher;
Plasma cells in human pancreatic ductal adenocarcinoma secrete antibodies to self-antigens
26 Sep 2023 | JCI insight | :e172449
Yao, Min; Preall, Jonathan; Yeh, Johannes; Pappin, Darryl; Cifani, Paolo; Zhao, Yixin; Shen, Sophia; Moresco, Philip; He, Brian; Patel, Hardik; Habowski, Amber; King, Daniel; Raphael, Kara; Rishi, Arvind; Sejpal, Divyesh; Weiss, Matthew; Tuveson, David; Fearon, Douglas;