Our immune systems have the potential to find and destroy cancer cells. But cancer cells can be clever and develop tricks to evade the immune system. Cold Spring Harbor Laboratory Professor Douglas Fearon and his former postdoc ZhiKai Wang found one such trick. Cancer cells weave a deactivating signal into a protective coat of armor that excludes T cells that would otherwise kill them. This immune deactivation pathway offers a promising new therapeutic approach for pancreatic, breast, and colorectal cancers.
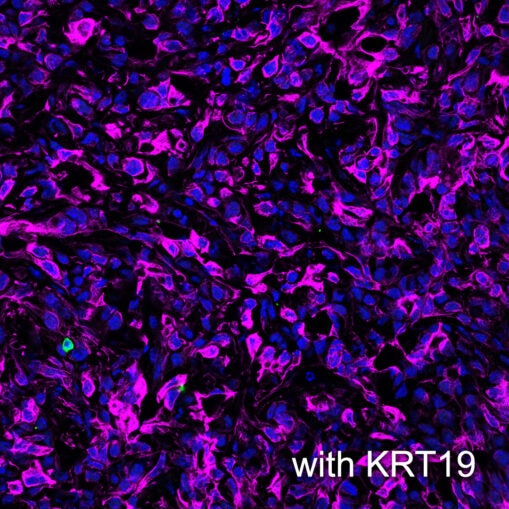
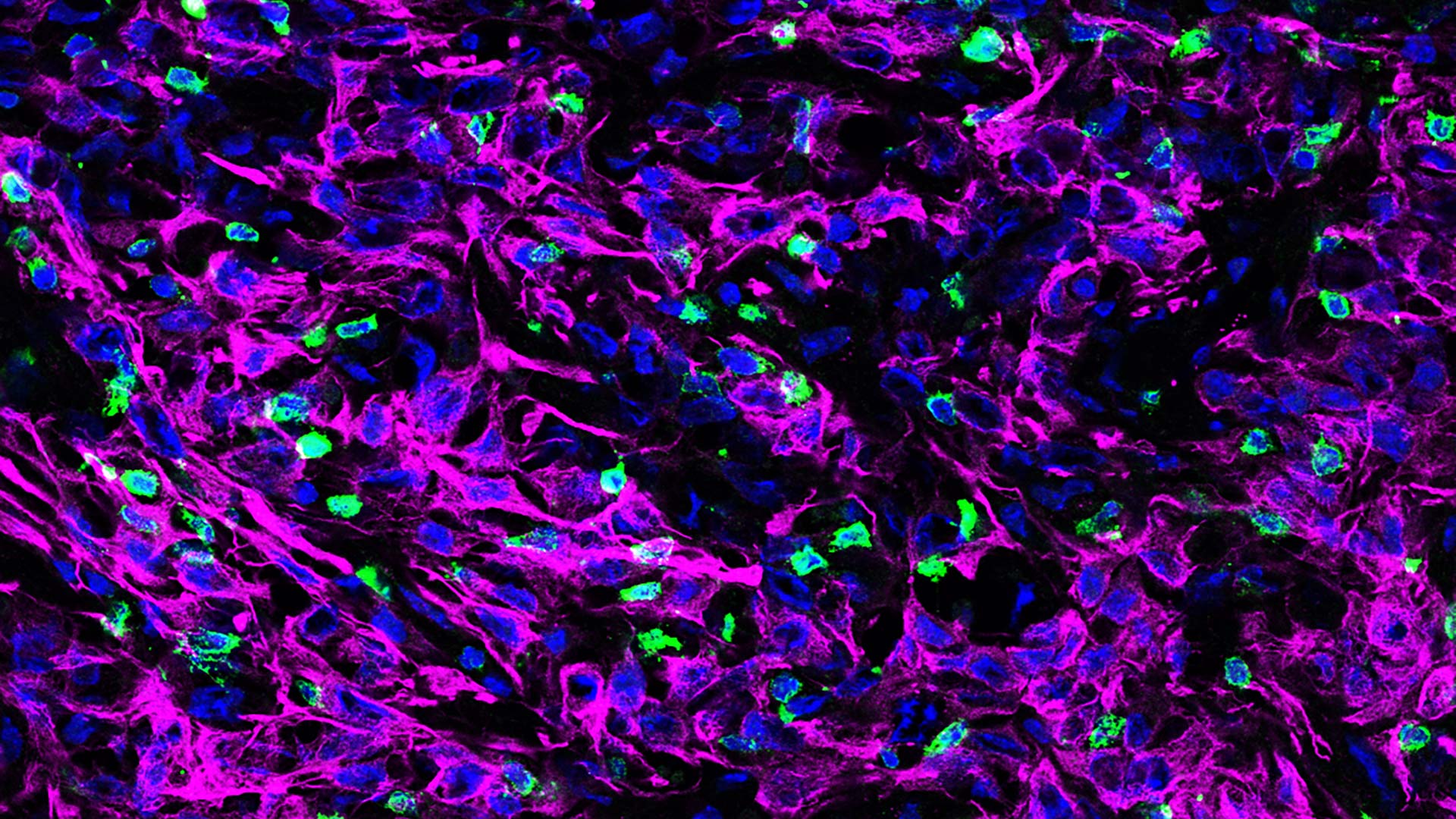
T cells patrol the body looking for cancers and pathogens. If they and/or their immune system teammates find an intruder, the T cells are mobilized to attack. Wang, currently a research fellow at the University of Science and Technology of China in Hefei, discovered this mobilization was disabled by a combination of three proteins woven into a protective coating surrounding cancer cells: a signal that usually attracts T cells called CXCL12, a filament called KRT19, and a protein that fuses the former proteins together called TGM2.
The scientists used genetic editing to turn off the production of KRT19 or TGM2 in mouse pancreatic tumors. Without KRT19 or TGM2, the cancer cells lost the CXCL12-KRT19 protection and T cells were able to infiltrate and attack. The pancreatic tumors shrank or disappeared.
Why did this coat of proteins repel T cells from the tumors? Wang says, “It is kind of counterintuitive because CXCL12 is a chemokine (chemical attractant) that attracts immune cells. But we found that CXCL12 is in an unusually high concentration on the surface of the cancer cells, where it does the opposite by making T cells immobile.” CXCL12 usually does its work as a single protein. But at high concentrations on the surface of cancer cells, the protein is in a complex with KRT19 and forms a branch-like network. T cell movement was reduced dramatically by this network.
The study was published in the Proceedings of the National Academies of Sciences. In a previous small clinical study of pancreatic cancer patients, Fearon and collaborators showed that the drug plerixafor (a CXCL12 receptor blocker) increased the infiltration of T cells into patients’ pancreatic tumor tissues. The current study now shows why this immunotherapeutic effect occurs. Fearon and Wang hope CXCL12 and KRT19 will provide new therapeutic targets that boost the immune system’s chances of killing off cancer cells.
A video showing how a component of cancer’s anti-immune armor (called the CXCL12/KRT19 complex) slows an important member of the immune system, called T cells, and stops them from entering a tumor. Video: ZhiKai Wang/Fearon lab
Written by: Luis Sandoval, Communications Specialist | sandova@cshl.edu | 516-367-6826
Funding
Lustgarten Foundation Distinguished Scholar Award, Thompson Family Foundation, Cedar Hill Foundation, National Cancer Institute, George A. & Marjorie H. Anderson Scholarship from the School of Biological Sciences, National Institutes of Health
Citation
Wang, Z., et al., “Carcinomas assemble a filamentous CXCL12–keratin-19 coating that suppresses T cell-mediated immune attack”, Proceedings of the National Academy of Sciences, January 25, 2022. DOI: 10.1073/pnas.2119463119
Principal Investigator

Douglas Fearon
Professor
Cancer Center Member
M.D., Johns Hopkins University School of Medicine, 1968