Business Development & Technology Transfer
CSHL is engaged in technology transfer as one way of delivering important discoveries to the public. Our Office of Technology Transfer actively supports our scientists to partner with companies, investors and others in order to achieve this mission and make a positive impact on society.
Guiding Principles
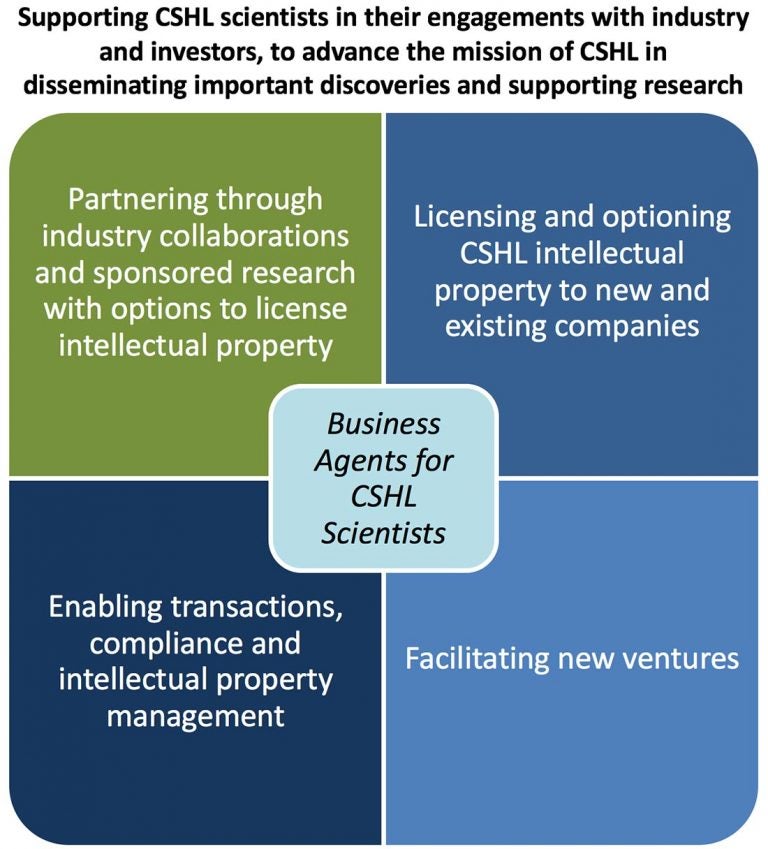
The purpose of CSHL’s technology transfer activities is to enable the scientific research conducted by our faculty, scientific staff and students to be developed and applied for the common good.To achieve this goal, we follow these guiding principles:
- Work collaboratively with CSHL scientists to support their aspirations, manage conflicts, comply with regulations and encourage synergy with CSHL’s mission
- Support effective dissemination of technology and leverage intellectual property rights and resources
- Encourage commercial opportunities with the potential to make a significant positive impact
- Engage with industry and investors to find the best partners to move ideas forward
- Negotiate fair and reasonable terms for deals that encourage commercialization, global access and good returns
Performance Metrics
The ultimate measure of our performance is the impact on society through our commercialization efforts. Early performance indicators include funds received, transactions entered into and relationships established and maintained.
Business Development & Technology Transfer News
- Cold Spring Harbor Laboratory and Deerfield Management Launch Harbor Discoveries to Advance New Therapeutics
- Envisagenics Announces Research Collaboration with Bristol Myers Squibb
- Veranome Biosystems and Cold Spring Harbor Laboratory Enter Collaboration and Licensing Agreement to Develop Advanced In-Situ Sequencing Technologies
- Envisagenics Raises Series A Financing to Scale Its AI-powered RNA Splicing Drug Discovery Platform
- Envisagenics to Bring RNA Splicing Software Platform to J&J Lung Cancer Initiative
- Envisagenics Awarded $300K NCI Grant to Refine Drug Discovery Tech
- uniQure Strengthens Intellectual Property Portfolio with Granted Patent Claims Covering AMT-130 for Huntington’s Disease
- New technology could predict cancer outcome
President’s essay: The continuous cycle of discovery
May 30, 2024
CSHL President & CEO Bruce Stillman discusses our institution’s societal impacts and global connections as forces for further scientific progress.
CSHL and Northwell Health extend strategic affiliation
April 29, 2024
Historic agreement aims to translate basic bioscience into a clinical setting, providing cancer patients greater access to personalized healthcare.
Bald eagles fight in flight
January 25, 2024
There’s been a surge of bald eagle sightings on campus. CSHL’s Vlad Drozdoff brings us an amazing close-up look at these birds in action.
Dream big: A powerful vision for CSHL research
January 2, 2024
New Cold Spring Harbor Laboratory Director of Research Leemor Joshua-Tor shares her vision for the future of bioscience discovery.
Breaking new ground: For science and society
November 13, 2023
CSHL’s Foundations for the Future campaign will propel the institution’s bioscience research and education programs to new heights and maximal impact.
Laying the groundwork for drug discoveries
August 8, 2023
A new partnership between CSHL and one of the world’s leading biotech investors could streamline this process and help change society for the better.
CSHL and Deerfield Management launch Harbor Discoveries
January 12, 2023
Deerfield commits up to $130 million over 10 years to support drug development efforts.
Envisagenics and Biogen partner for RNA splicing research
June 9, 2021
CSHL spin-out company Envisagenics teams up with Biogen to advance research in RNA-based therapeutics for central nervous system diseases.
New incubator model for CSHL spin-outs
June 7, 2021
When a CSHL lab discovers a potential drug target, Autobahn Labs will collaborate with us to direct CRO work at Evotec funded by Samsara BioCapital.
$11 million investment to develop fibroblast therapeutic
April 26, 2021
CSHL Professor David Tuveson and other leading disease experts founded a company to target fibroblasts as a new way to treat cancer and inflammatory diseases.
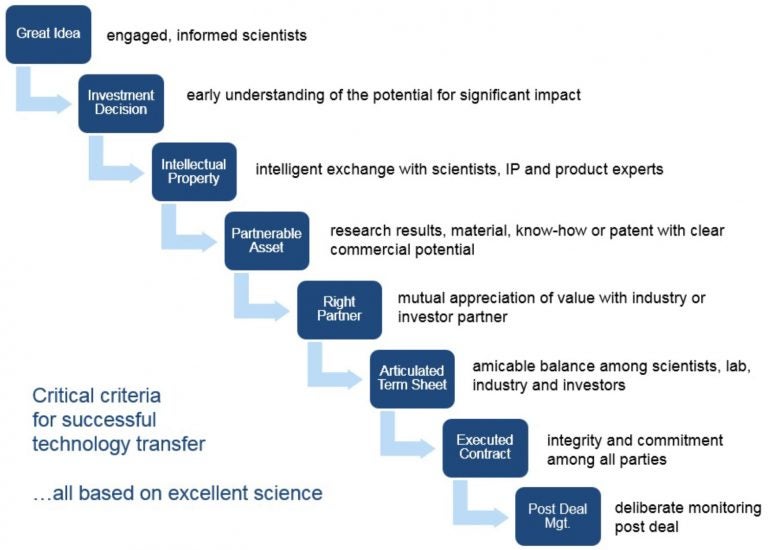
Technology Transfer Process
Our Scientist’s Guide outlines key elements of the technology transfer process at CSHL. Learn more.
Commercial Relations
Engagement with industry and commercial relationships are governed by CSHL’s Commercial Relations Policy. Learn more.
Technology Disclosure
If you are a CSHL scientist and think the results of your research have commercial potential, please come talk with us. We will discuss completing a Technology Disclosure Form and address any questions you have about our process. Learn more.
Material Transfer
The exchange of research materials is an essential part of scientific research. It promotes effective collaboration among CSHL scientists and their counterparts in academia, government and industry. Learn more.
Conflict of Interest Policy
The Investigator Conflict of Interest Policy sets forth specific procedures for annual disclosure and review of significant financial interests so that, as an institution, the highest standards of integrity and objectivity are applied to the design, conduct and reporting of research carried out at or by CSHL. Learn more.
Consulting
Consulting agreements are private contracts between investigators and companies outside the scope of CSHL employment. Investigators have an obligation to familiarize themselves with CSHL’s policies relevant to consulting activities before entering into any such arrangement. Learn more.
Faculty Guide
CSHL’s Faculty Guide provides an introduction to the areas of research interest for each member of our faculty.
Licensing
CSHL’s world-renowned research programs span Cancer, Neuroscience, Plant Biology, Quantitative Biology and Bioinformatics & Genomics. CSHL’s research environment is highly collaborative, allowing CSHL scientists to work together across disciplines to solve biology’s most challenging problems, with the ultimate goal of benefiting the public by advancing this research to improve the diagnosis and treatment of cancer, neurological disorders and other diseases. With that goal in mind, CSHL grants licenses to existing and startup companies that demonstrate the technical and financial capabilities to develop our early-stage technology into commercially successful products. Companies are encouraged to discuss their business interests with our Office of Technology Transfer so we can bring the CSHL inventions and prominent investigators in their areas of interest to their attention. Learn more.
Material Transfer Agreements
The Office of Technology Transfer supports CSHL scientists with agreements to transfer proprietary materials into and out of CSHL through Material Transfer Agreements as well as tangible material licensing arrangements.
shRNA Technology Licenses
CSHL research has played a major role in developing short hairpin RNAs (shRNAs) as important biomedical research tools that we wish to see distributed and used broadly. Learn more.
Sponsored Research
CSHL’s preferred sponsored research arrangement with industry would follow our Uniform SRA format.
In partnership with Hofstra University’s Zarb School of Business, Cold Spring Harbor Laboratory’s Business Development and Technology Transfer Office has developed a unique program aimed at providing students in Hofstra’s MBA program and CSHL’s School of Biological Sciences PhD program and postdocs with opportunities to gain insight and experiences in the mechanics of entrepreneurship within the life sciences industry.
Through both online and in-person experiences, the program is an immersive foundations of business experience where graduate students, law students and fellow scientists all learn what’s required to bring a vision to market. For more details on the program and application instructions, visit Hofstra’s Zarb School of Business program page here.
- Scientist Guide (pdf)
- Commercial Relations Policy (pdf)
- Invention Reporting Policy (pdf)
- Investigator Conflict of Interest Policy (pdf)
- Technology Evaluation Form (pdf)
- Technology Disclosure Form (pdf)
- Uniform Consulting Agreement Provisions (pdf)
- Uniform Sponsored Research Agreement
- CSHL’s Faculty Guide
- AUTM Material Transfer Agreement Template (pdf)
- AUTM Briefing Book: Key Issues in the Technology Transfer Business (pdf)
- Guide to Consulting Agreements for Biomedical Scientists
- NIH Genomic Data Sharing Policy
The Office of Technology Transfer is located in the Luke Building.
General contact information
Email: techtran@cshl.edu
Phone: 516-367-8301
Andrew Whiteley
Vice President
Business Development & Technology Transfer
whiteley@cshl.edu
Betsy Panagot
Executive Assistant & Office Manager
panagot@cshl.edu
Debra B. Arenare, J.D.
Vice President, General Counsel
darenare@cshl.edu
Vladimir Drozdoff, Ph.D., J.D.
Chief Patent Counsel
drozdoff@cshl.edu
Eric Greenbaum, J.D.
Director of New Ventures and AI
greenbaum@cshl.edu
Elizabeth A. Hand
Database, Accounting & Compliance Manager
hand@cshl.edu
Cynthia R. Miller, J.D.
Transactions Associate
miller@cshl.edu
Radhakrishnan (“RK”) Narayanan, Ph.D., M.B.A.
Senior Director
narayan@cshl.edu