Mikala Egeblad
Adjunct Professor
Ph.D., University of Copenhagen and the Danish Cancer Society, 2000
egeblad@cshl.edu | 516-367-6852
Cancer cells are surrounded by immune cells, blood vessels, chemical signals and a support matrix—collectively, the tumor microenvironment. Most microenvironments help tumors grow and metastasize, but some can restrict tumors. My lab studies how to target the bad microenvironments and support the good ones to combat cancer.
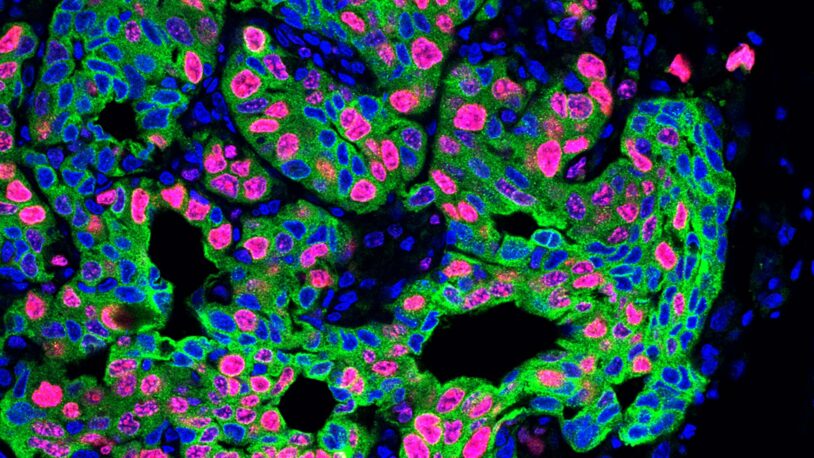
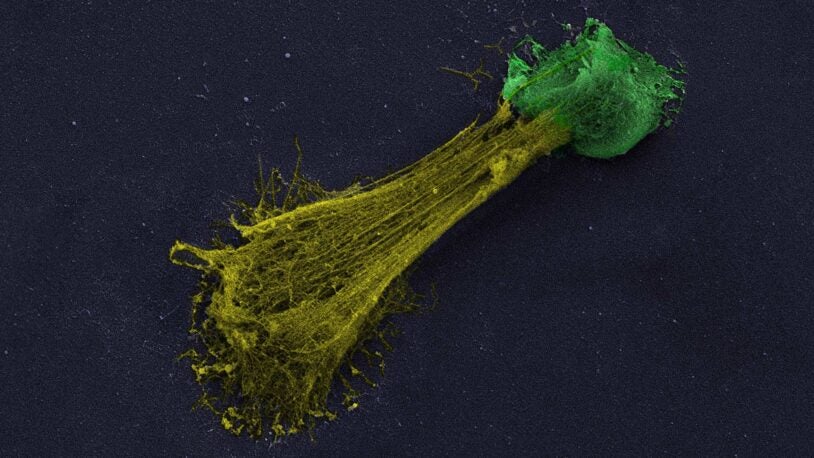
Mikala Egeblad and colleagues study cancer and, in particular, the microenvironment in which the cancer cells arise and live. Solid tumors are abnormally organized tissues that contain not only cancer cells, but also various other stromal cell types and an extracellular matrix, and these latter components constitute the microenvironment. Communications between the different components of the tumor influence its growth, its response to therapy, and its ability to metastasize. Among the tumor-associated stromal cells, the lab’s main focus is on myeloid-derived immune cells, a diverse group of cells that can enhance angiogenesis and metastasis and suppress the cytotoxic immune response against tumors. Egeblad is interested in how different types of myeloid cells are recruited to tumors and how their behaviors—for example, their physical interactions with cancer cells and other immune cells—influence cancer progression, including metastasis. The Egeblad lab studies the importance of the myeloid cells using mouse models of breast and pancreatic cancer and real-time imaging of cells in tumors in live mice. This enables them to follow the behaviors of and the interactions between cancer and myeloid cells in tumors during progression or treatment. This technique was instrumental when the lab showed that cancer drug therapy can be boosted by altering components of the tumor microenvironments, specifically reducing either matrix metalloproteinases (enzymes secreted by myeloid cells) or chemokine receptors (signal receptors on myeloid cells). Most recently, the Egeblad lab has showed that when a specific type of myeloid cell, called neutrophil, is activated during inflammation it can awaken sleeping cancer to cause cancer recurrence. The neutrophils do so by forming so-called neutrophil extracellular traps, structures of extracellular DNA and these alter the extracellular matrix surrounding the sleeping cancer cells to provide a wake-up signal.
The CSHL School of Biological Sciences’ class of 2025
May 5, 2025
The School of Biological Sciences awarded Ph.D. degrees to nine students this year. Read some of their stories and reflections on their time at CSHL.
At the Lab Season 1 Research Rewind: Cancer
October 8, 2024
As the first season of our new podcast winds down, we’re revisiting all of our episodes with a focus on CSHL’s cutting-edge cancer research.
At the Lab Episode 18: The stress of cancer
August 6, 2024
A sobering conversation on a breakthrough discovery with potentially significant implications for cancer patients everywhere.
Women’s health quiz
March 19, 2024
CSHL research has yielded insights into a number of women’s health topics, from menopause to breast cancer. Take this quiz to see how far we’ve come.
Chronic stress spreads cancer … here’s how
February 22, 2024
CSHL researchers have discovered a new link between chronic stress and cancer metastasis, providing a possible path forward for new treatments.
Dr. Egeblad receives support from the Katie Oppo Research Foundation
September 30, 2022
Dr. Mikala Egeblad receives a $60,000 grant from the Katie Oppo Research Foundation to support her work in metastatic ovarian cancer.
Drug disulfiram prevents immune-related damage
February 14, 2022
Severe lung infections (like SARS-CoV-2) can trigger a deadly cascade of immune events. In animal models, disulfiram can prevent a very toxic step.
Drug halts immune reactions to save damaged lungs
February 8, 2022
In two rodent studies, CSHL researchers found how disulfiram decreases immune-mediated damage after lung injury or infection with SARS-CoV-2.
Reprogramming the immune response in ovarian cancer
November 26, 2021
Swim Across America supports CSHL Professor Mikala Egeblad’s cancer research, including an open water fundraiser swimming event each summer.
Can our own immune system fight off cancer?
November 10, 2021
Researchers have studied the link between cancer and the immune system for centuries. Today, immunology provides promising solutions to combat cancer.
Selected Publications
Caught in a Web: Emerging Roles of Neutrophil Extracellular Traps in Cancer
11 Apr 2022 | Annual Review of Cancer Biology | 6(1)
He, Xue-Yan, Ng, David, Egeblad, Mikala
Disulfiram inhibits neutrophil extracellular trap formation protecting rodents from acute lung injury and SARS-CoV-2 infection.
8 Feb 2022 | JCI Insight
Adrover, Jose, Carrau, Lucia, Daßler-Plenker, Juliane, Bram, Yaron, Chandar, Vasuretha, Houghton, Sean, Redmond, David, Merrill, Joseph, Shevik, Margaret, tenOever, Benjamin, Lyons, Scott, Schwartz, Robert, Egeblad, Mikala
Innate Immunity and Cancer Pathophysiology
17 Nov 2021 | Annual Review of Pathology: Mechanisms of Disease | 17(1)
Maiorino, Laura, Daßler-Plenker, Juliane, Sun, Lijuan, Egeblad, Mikala
Activating a collaborative innate-adaptive immune response to control metastasis.
1 Sep 2021 | Cancer Cell
Sun, Lijuan, Kees, Tim, Almeida, Ana, Liu, Bodu, He, Xue-Yan, Ng, David, Han, Xiao, Spector, David, McNeish, Iain, Gimotty, Phyllis, Adams, Sylvia, Egeblad, Mikala
Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice
28 Sep 2018 | Science | 361(6409)
Albrengues, J, Shields, M, Ng, D, Park, C, Ambrico, A, Poindexter, M, Upadhyay, P, Uyeminami, D, Pommier, A, Kuttner, V, Bruzas, E, Maiorino, L, Bautista, C, Carmona, E, Gimotty, P, Fearon, D, Chang, K, Lyons, S, Pinkerton, K, Trotman, L, Goldberg, M, Yeh, J, Egeblad, M
Cancer cells induce metastasis-supporting neutrophil extracellular DNA traps
19 Oct 2016 | Science Translational Medicine | 8(361):361ra138
Park, J, Wysocki, R, Amoozgar, Z, Maiorino, L, Fein, M, Jorns, J, Schott, A, Kinugasa-Katayama, Y, Lee, Y, Won, N, Nakasone, E, Hearn, S, Kuttner, V, Qiu, J, Almeida, A, Perurena, N, Kessenbrock, K, Goldberg, M, Egeblad, M
All Publications
Tumour extracellular vesicles and particles induce liver metabolic dysfunction.
Jun 2023 | Nature | 618(7964):374-382
Wang, Gang, Li, Jianlong, Bojmar, Linda, Chen, Haiyan, Li, Zhong, Tobias, Gabriel, Hu, Mengying, Homan, Edwin, Lucotti, Serena, Zhao, Fengbo, Posada, Valentina, Oxley, Peter, Cioffi, Michele, Kim, Han, Wang, Huajuan, Lauritzen, Pernille, Boudreau, Nancy, Shi, Zhanjun, Burd, Christin, Zippin, Jonathan, Lo, James, Pitt, Geoffrey, Hernandez, Jonathan, Zambirinis, Constantinos, Hollingsworth, Michael, Grandgenett, Paul, Jain, Maneesh, Batra, Surinder, DiMaio, Dominick, Grem, Jean, Klute, Kelsey, Trippett, Tanya, Egeblad, Mikala, Paul, Doru, Bromberg, Jacqueline, Kelsen, David, Rajasekhar, Vinagolu, Healey, John, Matei, Irina, Jarnagin, William, Schwartz, Robert, Zhang, Haiying, Lyden, David
Neutrophil extracellular traps formed during chemotherapy confer treatment resistance via TGF-β activation
10 Apr 2023 | Cancer Cell | 41(4):757-775.e10
Mousset, Alexandra, Lecorgne, Enora, Bourget, Isabelle, Lopez, Pascal, Jenovai, Kitti, Cherfils-Vicini, Julien, Dominici, Chloé, Rios, Géraldine, Girard-Riboulleau, Cédric, Liu, Bodu, Spector, David, Ehmsen, Sidse, Renault, Shufang, Hego, Caroline, Mechta-Grigoriou, Fatima, Bidard, François-Clément, Terp, Mikkel, Egeblad, Mikala, Gaggioli, Cédric, Albrengues, Jean
T cell immunotherapies engage neutrophils to eliminate tumor antigen escape variants.
30 Mar 2023 | Cell | 186(7):1432-1447.e17
Hirschhorn, Daniel, Budhu, Sadna, Kraehenbuehl, Lukas, Gigoux, Mathieu, Schröder, David, Chow, Andrew, Ricca, Jacob, Gasmi, Billel, De Henau, Olivier, Mangarin, Levi, Li, Yanyun, Hamadene, Linda, Flamar, Anne-Laure, Choi, Hyejin, Cortez, Czrina, Liu, Cailian, Holland, Aliya, Schad, Sara, Schulze, Isabell, Betof Warner, Allison, Hollmann, Travis, Arora, Arshi, Panageas, Katherine, Rizzuto, Gabrielle, Duhen, Rebekka, Weinberg, Andrew, Spencer, Christine, Ng, David, He, Xue-Yan, Albrengues, Jean, Redmond, David, Egeblad, Mikala, Wolchok, Jedd, Merghoub, Taha
NETworking with cancer: The bidirectional interplay between cancer and neutrophil extracellular traps
18 Feb 2023 | Cancer Cell
Adrover, Jose, McDowell, Sheri, He, Xue-Yan, Quail, Daniela, Egeblad, Mikala