When confronted with another individual, social animals use multiple sensory inputs smells, sounds, sights, tastes, touches to choose an appropriate behavioral response. My group studies how specific brain circuits support these natural communication behaviors and how disruptions in these circuits can lead to inappropriate use of social information, as in Autism Spectrum Disorders.
Stephen Shea’s lab studies the neural circuitry underlying social communication and decisions. He uses natural social communication behavior in mice as a model to understand circuits and processes that are evolutionarily conserved and therefore shared broadly across species, likely contributing to disorders such as autism. Shea and colleagues have examined how emotion and arousal enable mice, via their olfactory systems, to store memories of other individuals and of related social signals. The team has exploited the intimate relationship between memory and emotion to effectively create memories in anesthetized mice, allowing them unprecedented access to neurobiological processes that typically only occur during behavior. The lab has been making a detailed analysis of the changes in neural connections that underlie odor memory. The team is particularly focused on an enigmatic cell type (granule cells or GCs) that has long been hypothesized to be crucial for memories, but has resisted direct study. They have developed methods for recording, giving them the first glimpse of the dynamics of these cells while the animal is learning an odor. The results show unexpectedly complex population dynamics among the GCs that were independently predicted by a model of odor learning developed in Alexei Koulakov’s lab. The two labs are collaborating to discern how GC population activity gets integrated by olfactory bulb output neurons and to pinpoint the synaptic circuit that underlies this form of learning. In parallel, another member of the lab is using imaging techniques to determine how memories are stored among broad neuronal ensembles, at a different level of the system. Recently, the lab made a key breakthrough, developing the ability to record from GCs in awake animals and discovering that their activity is dramatically modulated by state of consciousness. Finally, the Shea lab completed a series of studies of a different form of social recognition: auditory recognition of pup vocalizations by their mothers. Through this research, they have shown that a mouse model of Rett syndrome exhibits deficits in communication and learning not unlike those in human patients. Grants from the Simons and Whitehall Foundations are allowing the lab to extend this work by directly linking these deficits to the action of the gene MeCP2 in the auditory cortex.
A perception quiz to blow your mind
March 26, 2025
Ready to think differently about how you see, smell, hear, taste, touch, and make sense of the world around you?
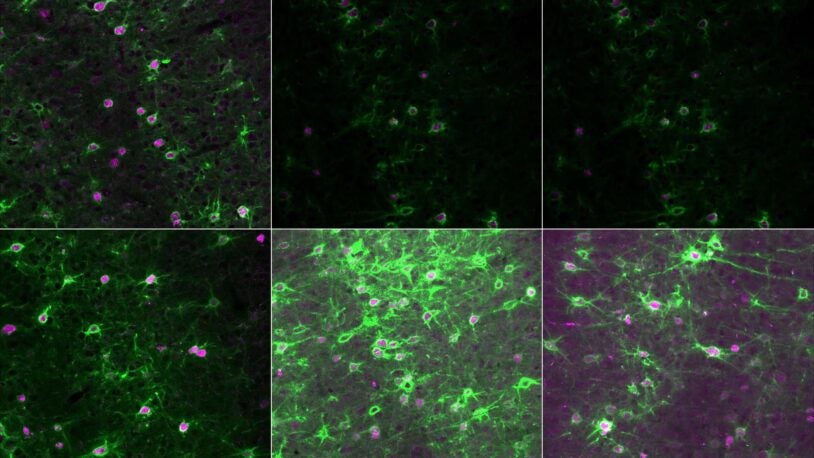
Mixed signals: How the brain interprets social cues
January 6, 2025
A new discovery from CSHL’s Shea lab helps explain how the brain processes multisensory information. It may have big implications for autism research.
At the Lab Season 1 Research Rewind: Neuroscience
October 15, 2024
What do you think? How do you know? And who are you anyway? We probe each of these questions with the help of Cold Spring Harbor’s neuroscientists.
Cocktails & Chromosomes: Of mice and moms
June 13, 2024
The latest installment in our public science talk series fell right between Mother’s Day and Father’s Day. CSHL’s Stephen Shea took it from there.
At the Lab Episode 6: Supermoms to the rescue
May 7, 2024
Happy almost Mother’s Day! What drives a supermom to come to the rescue when her child is upset? CSHL’s Stephen Shea shares the biological backstory.
Animal behavior quiz
December 20, 2023
Take this short quiz to see how much you know about the science of social behavior.
Reactivate, repurpose, and rewire the brain
October 4, 2023
New CSHL research on Rett syndrome could also have implications for autism spectrum disorders, dementia, and Alzheimer’s disease.
CSHL celebrates 20th graduating class
May 9, 2023
Friends, family, and faculty applauded 11 Ph.D. recipients at the CSHL School of Biological Sciences’ 2023 commencement.
The CSHL School of Biological Sciences’ class of 2023
May 7, 2023
The School of Biological Sciences awarded 11 Ph.D. degrees this year. Here, the graduates reflect on their time and experiences at CSHL.
What drives mom to pick up the kids?
February 9, 2023
The relationship between a mother and child is special. This video shows how moms’ brains are wired to reward motherly care.
All Publications
Multisensory integration of social signals by a pathway from the basal amygdala to the auditory cortex in maternal mice
26 Nov 2024 | Current Biology
Nowlan, Alexandra; Choe, Jane; Tromblee, Hoda; Kelahan, Clancy; Hellevik, Karin; Shea, Stephen;
Diversity of ancestral brainstem noradrenergic neurons across species and multiple biological factors
16 Oct 2024 | bioRxiv
Kelberman, Michael; Rodberg, Ellen; Arabzadeh, Ehsan; Bair-Marshall, Chloe; Berridge, Craig; Berrocoso, Esther; Breton-Provencher, Vincent; Chandler, Daniel; Che, Alicia; Davy, Oscar; Devilbiss, David; Downs, Anthony; Drummond, Gabrielle; Dvorkin, Roman; Fazlali, Zeinab; Froemke, Robert; Glennon, Erin; Gold, Joshua; Ito, Hiroki; Jiang, Xiaolong; Johansen, Joshua; Kaye, Alfred; Kim, Jenny; Kuo, Chao-Cheng; Liu, Rong-Jian; Liu, Yang; Llorca-Torralba, Meritxell; McCall, Jordan; McElligott, Zoe; McKinney, Andrew; Miguelez, Cristina; Min, Ming-Yuan; Nowlan, Alexandra; Omrani, Mohsen; Poe, Gina; Pickering, Anthony; Ranjbar-Slamloo, Yadollah; Razquin, Jone; Rodenkirch, Charles; Sales, Anna; Satyasambit, Rath; Shea, Stephen; Sur, Mriganka; Tkaczynski, John; Torres-Sanchez, Sonia; Uematsu, Akira; Vazquez, Chayla; Vreven, Amelien; Wang, Qi; Waterhouse, Barry; Yang, Hsiu-Wen; Yang, Jen-Hau; Zhao, Liping; Zouridis, Ioannis; Weinshenker, David; Vazey, Elena; Totah, Nelson;
Selective deletion of Methyl CpG binding protein 2 from parvalbumin interneurons in the auditory cortex delays the onset of maternal retrieval in mice
24 Aug 2023 | Journal of Neuroscience | :JN-RM
Rupert, Deborah; Pagliaro, Alexa; Choe, Jane; Shea, Stephen;
A circuit from the locus coeruleus to the anterior cingulate cortex modulates offspring interactions in mice
25 Jul 2023 | Cell Reports | 42(7):112771
Corona, Alberto; Choe, Jane; Muñoz-Castañeda, Rodrigo; Osten, Pavel; Shea, Stephen;
Selective deletion of Methyl CpG binding protein 2 from parvalbumin interneurons in the auditory cortex delays the onset of maternal retrieval in mice.
31 Jan 2023 | bioRxiv
Rupert, Deborah; Pagliaro, Alexa; Choe, Jane; Shea, Stephen;
A dopaminergic reward prediction error signal shapes maternal behavior in mice
13 Dec 2022 | Neuron | :S0896-6273(22)01073
Xie, Yunyao; Huang, Longwen; Corona, Alberto; Pagliaro, Alexa; Shea, Stephen;
A circuit from the locus coeruleus to the anterior cingulate cortex modulates offspring interactions in mice
2022 | bioRxiv
Corona, Alberto; Choe, Jane; Muñoz-Castañeda, Rodrigo; Osten, Pavel; Shea, Stephen;
Neurotensin neurons in the extended amygdala control dietary choice and energy homeostasis
20 Oct 2022 | Nature Neuroscience
Furlan, Alessandro; Corona, Alberto; Boyle, Sara; Sharma, Radhashree; Rubino, Rachel; Habel, Jill; Gablenz, Eva; Giovanniello, Jacqueline; Beyaz, Semir; Janowitz, Tobias; Shea, Stephen; Li, Bo;
The Bruce effect: Representational stability and memory formation in the accessory olfactory bulb of the female mouse
23 Aug 2022 | Cell Reports | 40(8):111262
Yoles-Frenkel, Michal; Shea, Stephen; Davison, Ian; Ben-Shaul, Yoram;
Parvalbumin-Positive Interneurons Regulate Cortical Sensory Plasticity in Adulthood and Development Through Shared Mechanisms
6 May 2022 | Frontiers in Neural Circuits | 16:886629
Rupert, Deborah; Shea, Stephen;