When organisms pass their genes on to future generations, they include more than the code spelled out in DNA. Some also pass along chemical markers that instruct cells how to use that code. The passage of these markers to future generations is known as epigenetic inheritance. It’s particularly common in plants. So, significant findings here may have implications for agriculture, food supplies, and the environment.
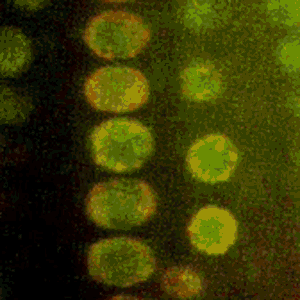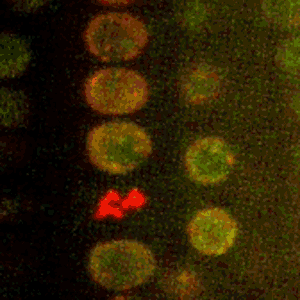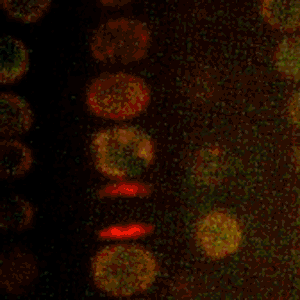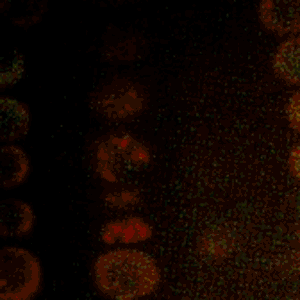
Cold Spring Harbor Laboratory (CSHL) Professors and HHMI Investigators Rob Martienssen and Leemor Joshua-Tor have been researching how plants pass along the markers that keep transposons inactive. Transposons are also known as jumping genes. When switched on, they can move around and disrupt other genes. To silence them and protect the genome, cells add regulatory marks to specific DNA sites. This process is called methylation.
Martienssen and Joshua-Tor have now shown how protein DDM1 makes way for the enzyme that places these marks on new DNA strands. Plant cells need DDM1 because their DNA is tightly packaged. To keep their genomes compact and orderly, cells wrap their DNA around packing proteins called histones. “But that blocks access to the DNA for all sorts of important enzymes,” Martienssen explains. Before methylation can occur, “you have to remove or slide the histones out of the way.”
Martienssen and former CSHL colleague Eric Richards first discovered DDM1 30 years ago. Since then, researchers have learned it slides DNA along its packing proteins to expose sites needing methylation. Martienssen likens the movement to a yo-yo gliding along a string. The histones “can move up and down the DNA, exposing parts of the DNA at a time, but never falling off,” he explains.
Through genetic and biochemical experiments, Martienssen pinpointed the exact histones DDM1 displaces. Joshua-Tor used cryo-electron microscopy to capture detailed images of the enzyme interacting with DNA and associated packing proteins. They were able to see how DDM1 grabs onto particular histones to remodel packaged DNA. “An unexpected bond that ties DDM1 together turned out to correspond to the first mutation found all those years ago,” Joshua-Tor says.
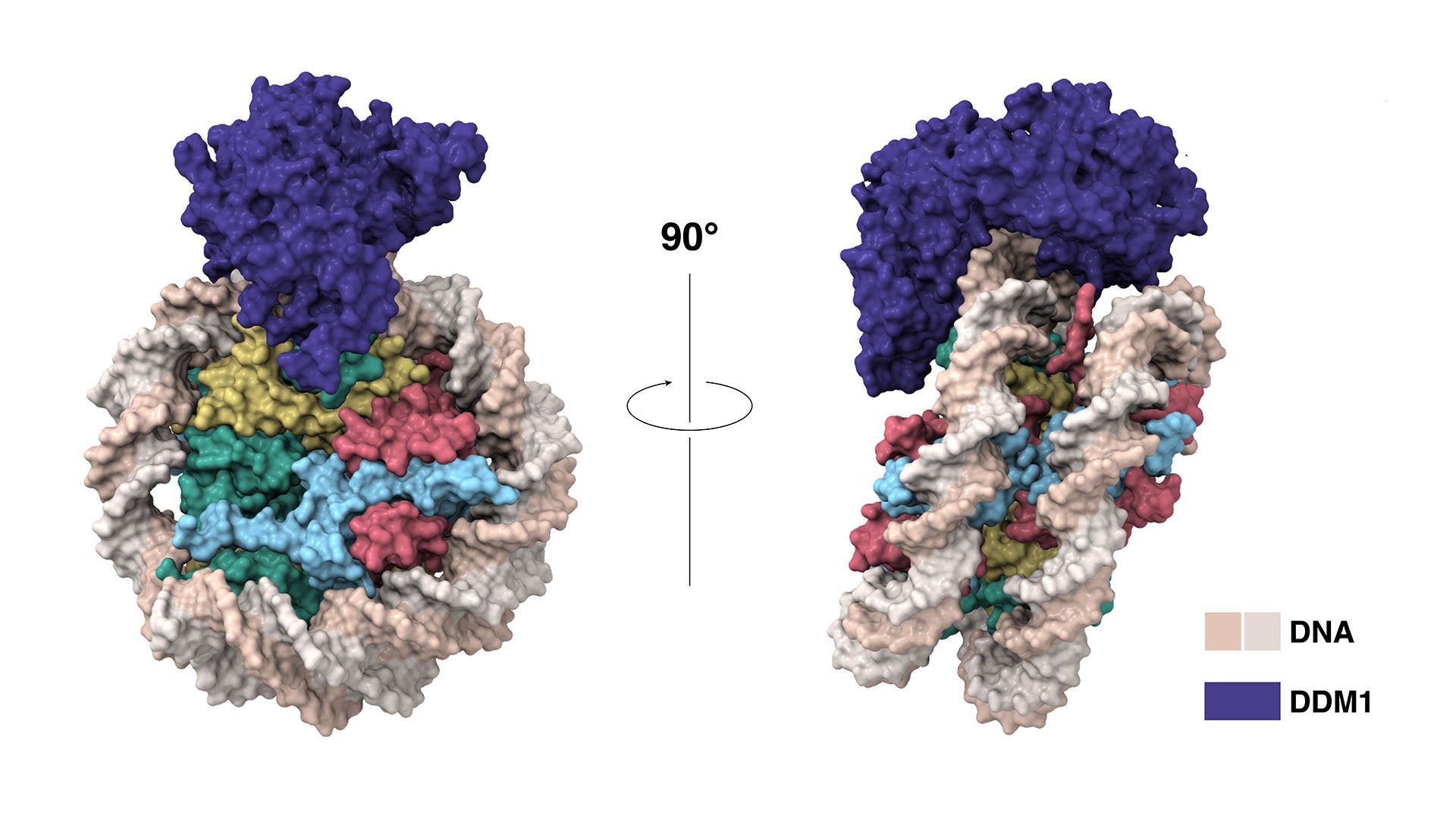
The experiments also revealed how DDM1’s affinity for certain histones preserves epigenetic controls across generations. The team showed that a histone found only in pollen is resistant to DDM1 and acts as a placeholder during cell division. “It remembers where the histone was during plant development and retains that memory into the next generation,” Martienssen says.
Plants may not be alone here. Humans also depend on DDM1-like proteins to maintain DNA methylation. The new discovery may help explain how those proteins keep our genomes functional and intact.
Written by: Jennifer Michalowski, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
National Institutes of Health, National Science Foundation Plant Genome Research Program, Howard Hughes Medical Institute, Wellcome Trust, European Research Council, Horizon 2020.
Citation
Lee, S.C., et al., “Chromatin remodeling of histone H3 variants underlies epigenetic inheritance of DNA methylation”, Cell, August 28, 2023. DOI: 10.1016/j.cell.2023.08.001

