Matthew Weiss dreams of the day when his oncology practice will operate very differently. A surgeon at the Northwell Health System in New York who treats pancreatic cancer—one of the deadliest malignancies known—he doesn’t have a lot of choices when it comes to saving his patients. Some people with pancreatic tumors die within a few weeks and others fight longer, but only 11% of them are still alive five years later. Current treatment options are limited. There are only two treatment paradigms, one based on a cocktail of two chemotherapy drugs and another one based on three, but doctors never know which one will work. “We may as well flip a coin when we decide which regimen to use,” says Weiss. “We have no way to predict who’s going to respond to what chemotherapy.”
Doctors can of course switch from one regimen to another if the initial performs poorly. But it takes a few months to determine whether tumors are shrinking or not, and patients don’t have that time. Moreover, the initial chemo may sicken some to the point that they are too weak to try the second approach. To make things worse, the incidences of pancreatic cancer, once considered rare, have increased in the past 20 years, now reaching over 60,000 cases annually in America alone. “About 1 in 50 to 60 people get it, and most of them don’t survive it,” says David Tuveson, director of Cold Spring Harbor Laboratory’s Cancer Center and director of research for the Lustgarten Foundation. “It used to be rare, but it’s not anymore. It is now the third most common cancer, behind lung and colon, and predictions show that it’s going to take the number two spot soon.”
Even more concerning is the fact that pancreatic cancer now strikes early, afflicting a greater number of young people, particularly women under 35. “I saw a patient yesterday morning who is 41 years old, with pancreatic cancer and three young children at home,” Weiss shares. With an average prognosis of about five years to live, it’s a family tragedy. “Five years is not enough, so to me pancreatic cancer is an emergency,” Weiss says. “We have to do things radically different.” Treatment must take guessing out of the process.
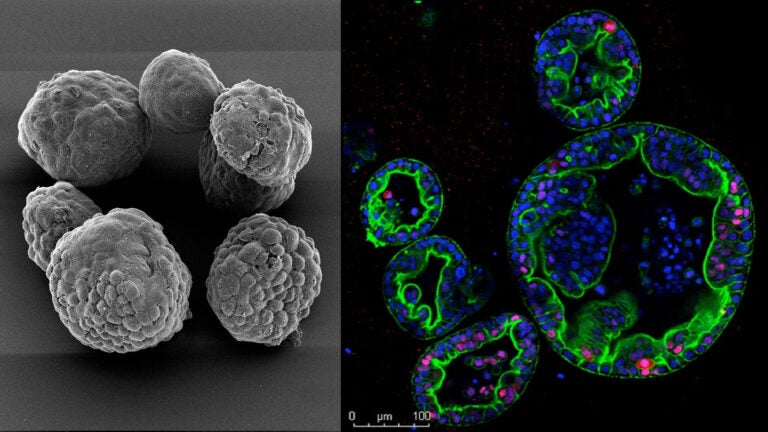
Weiss dreams of having a way to test the regimens before placing people on them. For the past few years, in collaboration with Tuveson, he has been working on making this a reality. Their method involves trying the chemo cocktails on pancreatic tumors outside the patient who developed them. In scientific terms, such outside tumors are called organoids. They’re grown from the small chunks of cancer cells biopsied from patients as part of their diagnosis. Because they retain the characteristics and mutations of the original tumor, their behavior when grown in a lab may predict their behavior inside their human host, including their reaction to the two chemo cocktails oncologists use to treat pancreatic cancer.
If the method works, it will improve patients’ current survival chances. But the idea has far-reaching potential. It may revolutionize how doctors treat cancers in the 21st century.

Cancers are tricky. Patients respond differently to the same treatments. One beats cancer and the other dies quickly. To battle cancer better, the treatment should be tailored to patients’ individual needs—the personalized medicine concept that has taken a center stage in cancer care. Organoids, combined with the genomic data from patients, might provide that much-needed personalized care. These little living tumors can also inform scientists how cancer develops, what changes it causes in the body in early and late stages, and how it plays out in people of different ethnic origins. That can shift not only the treatment approaches, but the entire cancer care paradigm.
In February 2022, President Joe Biden rekindled the Cancer Moonshot effort, which aims to end cancer as we know it. The initiative sets several goals, including reducing death rates by at least half in the next 25 years while improving the quality of life for those battling the disease. Currently, cancer kills about 600,000 Americans a year, and cutting it in half is a multi-pronged challenge. “The White House announcement makes you think, ‘What do we need to do to achieve that?’” Tuveson says. He sees the three pillars of success as better prevention, better detection, and better treatment.
Patient-derived organoids can be used to quickly and accurately predict how pancreatic cancer patients will respond to a variety of treatments, facilitating a precision-medicine approach to the deadly disease.
Better prevention begins with understanding cancer triggers, which in the case of the pancreas can begin with the complex interplay of the digestive organs. Your stomach digests food and absorbs the nutrients needed to sustain the body, but it can’t do all this hard work alone. It does it in concert with the other neighboring organs, including the pancreas and the liver. The banana-shaped pancreas produces the hormones insulin and glucagon, which regulate blood sugar levels. It also makes the digestive enzymes amylase, protease, and lipase, which respectively break down starches, proteins, and fats. Once made, these enzymes pour through the pancreatic ducts and into the upper part of the small intestine, called the duodenum, where they help digest food. The liver aids with fat breakdown by producing bile, which accumulates in the gallbladder, and then trickles into the stomach via bile ducts. “Bile is like soap, a green, greasy material that dissolves fat,” explains Tuveson.
This normally well-oiled machinery can get thrown off track by inflammatory processes. One of them is pancreatitis—an inflammation of pancreatic tissues, which can be caused by solidified bile, more commonly called gallbladder stones. “Sometimes when a stone passes from the gallbladder into the intestines, it may go the wrong way and get stuck in the pancreatic ducts, making the pancreas swell like a balloon,” Tuveson explains. “Now the enzymes can’t get out, so they start digesting the pancreas itself. And we don’t know why, but almost everybody who develops pancreatic cancer has had either symptomatic or silent pancreatitis before it happens.” Other things like alcohol overuse, smoking, obesity, and certain medicines can trigger pancreatitis also, which can increase cancer risk. “Many more patients develop pancreatitis than pancreatic cancer, so if we can prevent pancreatitis, we will probably also decrease the number of people who develop pancreatic cancer,” Tuveson says.
Organoids might help decipher the differences in cancer behavior between various ethnic groups, says Jeff Boyd, who directs the Northwell Health Cancer Institute’s Center for Genomic Medicine, while also holding a professorship at Cold Spring Harbor Laboratory. For example, pancreatic cancer outcomes are worse for African American and Latino people compared to those of European descent. It could be because of diets, pollution, or lifestyle—or due to some biological differences in the pancreatic cells. Organoids may hold the answers. “We’ve been trying to study the disparity of pancreatic cancer by culturing pancreas cancer organoids from individuals of European ancestry, African ancestry, and Hispanic ancestry,” Tuveson says. “So that we could investigate if there’s any fundamental difference in the biology of tumors from patients of different racial and ethnic backgrounds.”
Better detection requires better imaging procedures or specialized blood tests, neither of which clinicians have right now. Scientists know that an intraductal papillary mucinous neoplasm (IMPN)—a tiny growth arising from the duct lining—can be a precursor for pancreatic cancer, but spotting them early is difficult. Hidden behind the intestines, the pancreas is very hard to image, even with modern MRI and CT machinery. It doesn’t help that pancreatic tumors are usually microscopically small, compared to, for example, an intestinal polyp that gastroenterologists can identify with the naked eye. “An early pancreas tumor is the size of a grain of rice or even smaller,” Tuveson says, so they are easy to miss on a digital image. Inserting a camera scope into the pancreas through its very narrow ducts is an impossible feat because it would stop the flow of enzymes, which would then immediately start digesting the pancreas’ own tissues. A smarter way would be to develop a blood test that checks for certain compounds which a precancerous pancreas may produce, but so far, scientists don’t know of any.
That’s another problem Tuveson is trying to solve with organoids. His team grows IPMN organoids of these “early cancers” and implants them into the pancreatic ducts of mice, trying to see if this would spark any changes in the animals’ blood composition. Tuveson is also using mice implanted with IPMN cells to test new ways to image tiny tumors. “We’re growing early human cancers inside the duct of a mouse pancreas, so that we can find new ways to look for evidence in blood or develop new ways to image the cancer,” he says. “We use organoids in these futuristic kinds of approaches as a way to develop early detection methods.”
Better treatment is where organoids really get their chance to shine. And to some extent they are shining already, as a part of a bigger multi-institute and international effort—a clinical trial called PASS-01, which stands for Pancreatic Adenocarcinoma Signature Stratification for Treatment. When Weiss takes a biopsy, he sends the little cancerous bits to Tuveson’s laboratory where they are grown into organoids in Petri dishes. Then, Tuveson’s lab subjects the patient’s organoids to both chemo regimens, noting which one kills cancer more efficiently. Meanwhile, Weiss’s colleagues treat the patient on the chemo regimens. After a while, they compare results and see whether the patients’ tumors and the organoids reacted to treatment the same way. The team hopes to use this clinical trial to show the first scientific proof that organoids can indeed predict how tumors will respond inside the human body.
At this early clinical stage, the two collaborators aren’t using the organoids to decide which treatment to prescribe—they are only trying to establish the correlation. But so far, the results are encouraging. “We have a series of anecdotes that are promising,” Tuveson says. “And when you line up a bunch of anecdotes, you suddenly have a trend.” When the trend continues, and there are enough cases to undergo rigorous statistical analysis, one now has a case for the next level of a clinical trial: Using organoids to predict the best therapy and then treating the patient with it.
Another gift that organoids would give clinicians is the flexibility to experiment. With cancers in a dish, doctors can experiment all they want. They can alter drug ratios, such as decreasing the dose of a more toxic drug and see if the cocktail works just the same. They can swap one drug for another, including those not normally used in the current regimens, and see if it works better. And they can add another medication that’s not typically used for pancreatic cancer at all—and see if that does any miracles.
Tuveson likens the approach to shooting arrows at the enemy and finally finding its most vulnerable spot. “What we are doing is essentially looking for the cancer’s Achilles heel,” he says. And with the ability to test all kinds of approaches on organoids grown in a dish, there’s no limit to the number of arrows scientists can shoot. Oncologists will be able to do all the trial and error in a dish, sparing their patients from the chemo side effects, lost time, and diminishing recovery chances.
Weiss and Tuveson hope to see the decades-old treatment concepts change within a few years. “Treating cancer should work similar to treating infections,” Tuveson says. If you have a bad infection, the doctors may prescribe broad-spectrum antibiotics at first, but at the same time they also send your blood and urine tests to the lab and wait to see what bug you have. Once they know the exact culprit, they often change the meds to the most efficient antibiotic against it. “I’d like for the organoids to do the same—use them as a kind of a bacteriology test for cancer that will tell us the best way to kill it,” Tuveson says. “I’m optimistic that they can point us to cancer’s Achilles heel.”
Written by: Lina Zeldovich, Science Writer | publicaffairs@cshl.edu | 516-367-8455