Ketamine has received a Hollywood makeover. It used to be known as a rave drug (street name special K) and cat anesthetic. However, in recent years, some doctors have prescribed ketamine to treat conditions from post-traumatic stress disorder to depression. “The practice is not without controversy,” notes Cold Spring Harbor Laboratory (CSHL) Professor Hiro Furukawa.
‘Should we give a hallucinogen to patients in compromised mental states?’ wonder ketamine’s skeptics. The controversy came to a head in 2024 following the death of Matthew Perry. The popular TV actor, best known as Chandler on NBC’s Friends, died from a ketamine overdose. One person charged in connection with Perry’s death was the doctor who’d prescribed him ketamine for depression and anxiety.
This 3D animation illustrates the tension-and-release mechanism that controls how brain receptor GluN1-2B-2D opens and closes its ion channel pore.
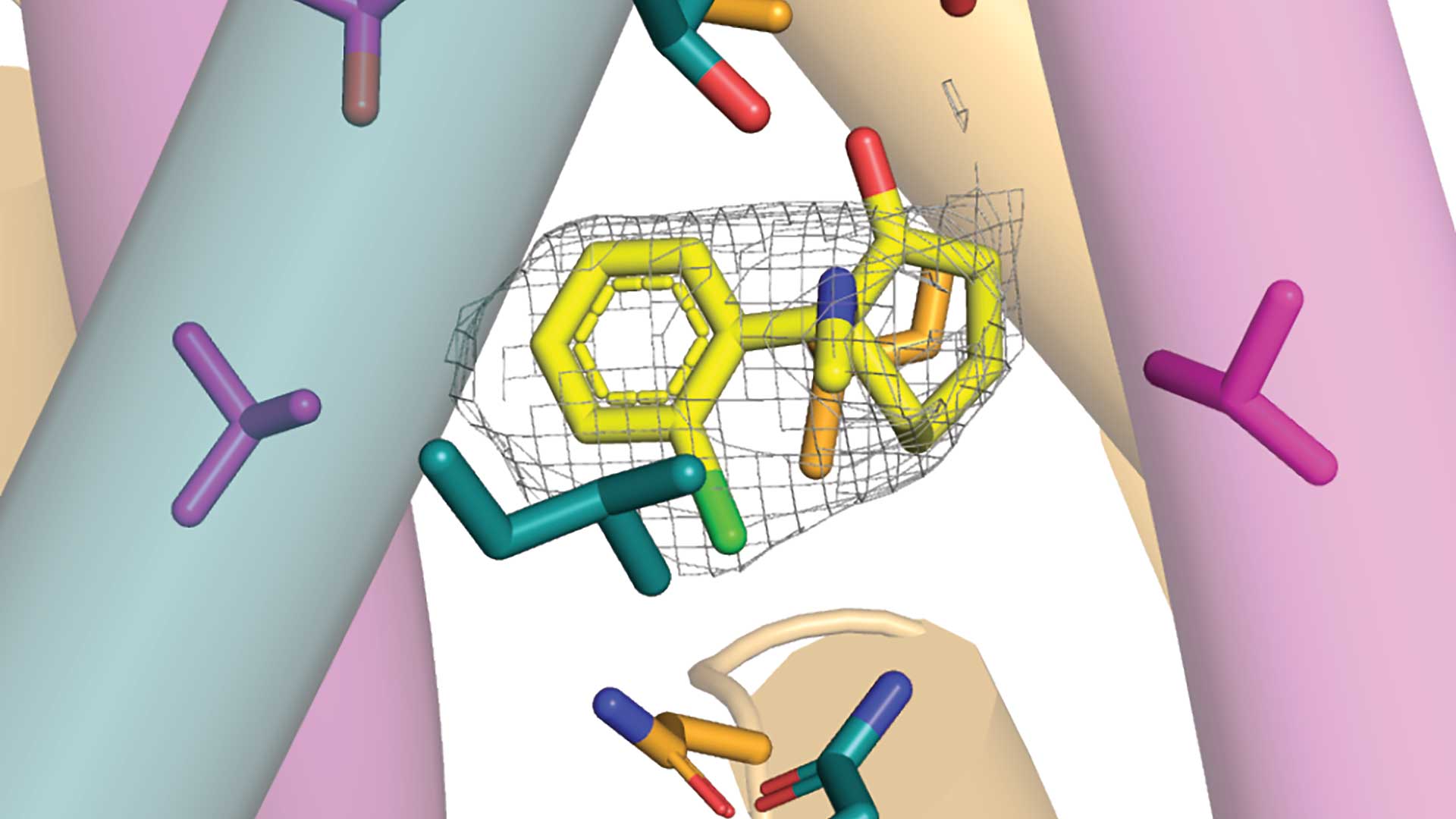
“Even putting this aside, many questions remain regarding how ketamine affects the brain,” says Furukawa. “It’s been suggested for over a decade that the drug blocks a specific kind of NMDA receptor (NMDAR), called GluN1-2B-2D.” There was one big problem with this theory. Scientists weren’t quite sure that GluN1-2B-2D existed. A new study from the Furukawa lab shines much-needed light on the situation.
In a paper published in the journal Neuron, Furukawa and postdoc Hyunook Kang prove that GluN1-2B-2D does exist in the mammal brain. They then reconstruct a human version of GluN1-2B-2D. They don’t stop there. Using electron cryo-microscopy (cryo-EM), they capture GluN1-2B-2D in action. The neuroscientists identify the tension-and-release mechanism that controls GluN1-2B-2D movements. They can now see how this mysterious NMDAR opens and closes its ion channel pore. And they go another step further. They reveal several ways ketamine may bind to GluN1-2B-2D.
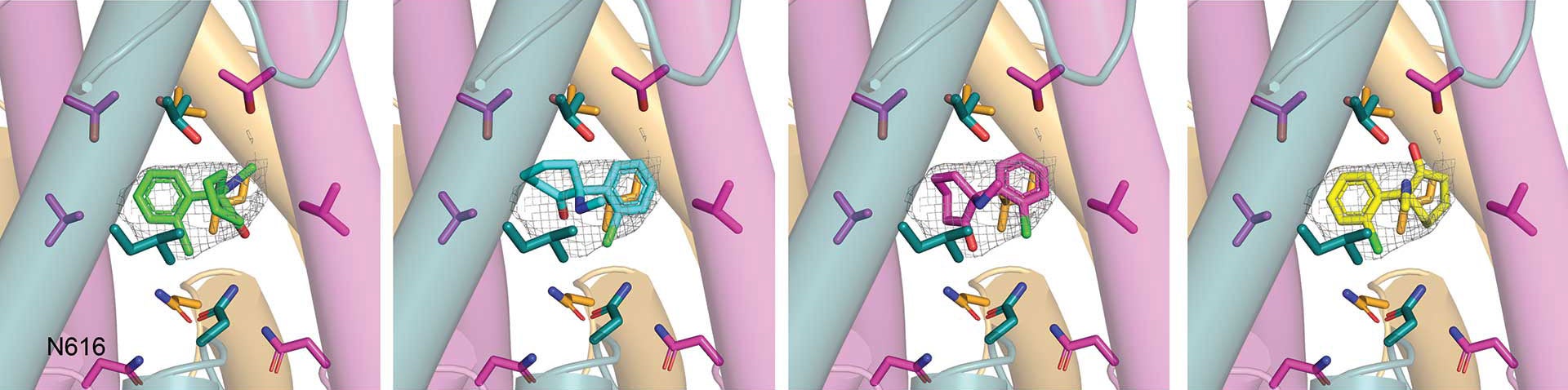
A series of stunningly detailed visualizations show ketamine molecules becoming attached to specific parts of GluN1-2B-2D. “It’s like a mesh,” explains Furukawa. “Over tiny fractions of a second, ketamine can latch onto these sections and close off the channel.” Furukawa and his colleagues captured four binding patterns. However, they believe there are many other ways ketamine can take hold.
It’s thought that ketamine may ease symptoms of depression and anxiety by affecting GluN1-2B-2D movement. But for how long should the channel remain open or closed? “This likely varies per patient,” Furukawa says. Likewise, side effects of ketamine therapy can range from mild hallucinations to full-on psychosis. However, if scientists can determine how GluN1-2B-2D movements affect the brain, they may be able to synthesize new versions of the drug with fewer harmful side effects. That could offer hope for millions of people living with depression and anxiety. So, that’s where Furukawa and his colleagues at CSHL will set their sights next.
Written by: Samuel Diamond, Editorial Content Manager | diamond@cshl.edu | 516-367-5055
Funding
National Institutes of Health, Austin’s Purpose, Robertson Research Fund, Doug Fox Alzheimer’s Fund, Heartfelt Wings Foundation, Gertrude and Louis Feil Family Trust, National Research Foundation of Korea
Citation
Kang, H., et al., “Structural basis for channel gating and blockade in tri-heteromeric GluN1-2B-2D NMDA receptor”, Neuron, February 14, 2025. DOI: 10.1016/j.neuron.2025.01.013
Core Facilites
Principal Investigator

Hiro Furukawa
Professor
Cancer Center Member
Ph.D., The University of Tokyo, 2001