It’s 2050, and you’re due for your monthly physical exam. Times have changed, so you no longer have to endure an orifices check, a needle in your vein, and a week of waiting for your blood test results. Instead, the nurse welcomes you with, “The doctor will sniff you now,” and takes you into an airtight chamber wired up to a massive computer. As you rest, the molecules you exhale or emit into the air slowly drift into the complex artificial intelligence (AI) apparatus, colloquially known as Deep Nose. Behind the scene, Deep Nose’s massive electronic brain starts crunching through the molecules, comparing them to its enormous olfactory database. Once it’s got a noseful, the AI matches your odors to medical conditions and generates a printout of your health. Your human doctor goes over the results with you and plans your treatment or adjusts your meds.

Odorprints
What can someone’s scent say about their health? Apparently, a lot. “The information that can be picked up from the airborne molecules is amazingly rich,” says Dmitry Rinberg, another former physicist and now a neurobiologist at New York University who collaborates with Koulakov on olfactory research. “It’s so informative that you can tell what kind of beer people drank at a bar last night,” he adds. “So we are trying to use this information for odor-based diagnostic approaches.”
The information that can be picked up from the airborne molecules is . . . so informative that you can tell what kind of beer people drank at a bar last night.”
Dmitry Rinberg, Ph.D.
Recent research has found that many diseases, including cancer, tuberculosis, and Parkinson’s, can manifest themselves through volatile compounds that change a person’s scent. Our bodies release many metabolites—products of our metabolic activities. Some of these molecules are volatile and become part of our scent, or “odorprint.” When we’re sick, these metabolic processes start functioning differently, emitting different molecules that change our odorprint. “These molecules carry information about our state of health,” Koulakov says. For example, patients with Parkinson’s disease produce an unusually high amount of sebum, a waxy lipid-rich biofluid excreted by the skin’s sebaceous glands, which sensitive noses can detect. Deep Nose could grab this type of information from the air. That could allow physicians to detect diseases sooner, easier, and perhaps avoid some invasive diagnostic procedures. “It would essentially revolutionize the diagnostics system,” Koulakov says.
Hippocrates, Galenus, Avicenna, and other physicians of ancient times used their noses as diagnostic tools. A wound with a nasty smell could mean it was infected. And bad breath signaled a host of ailments. Today, however, physicians don’t sniff their patients—because humans generally stink at smelling. In fact, we are worse than our ancestors. Our primate predecessors sported about 850 olfactory receptor types. But we only have 350 functional ones; the rest of them simply don’t work. “They are the remnants of our former glory,” Koulakov quips. Meanwhile, dogs have about 850 receptor types and mice about 1,100, so they are capable of discerning a much greater variety of smells—including those produced by the malfunctions of our bodies.
Pet doctors
Scientists now use that animal olfactory wealth to diagnose disease in peer-reviewed studies with some documented successes. Recently, a group of scientists from several research institutions reported that three trained beagles detected lung cancer cells in patient blood samples with 97% accuracy. In another recent study, dogs were able to detect colorectal cancer by smelling stool. A paper in the BioMed Central Cancer journal described dogs smelling out ovarian cancer. And in Sub-Saharan Africa, African giant pouched rats have been taught to work as “tuberculosis diagnosticians,” sniffing phlegm samples from patients.

This led scientists to ponder the possibility of an electronic nose instead. It would be far more economical to build an artificial sniffer apparatus that wouldn’t die after a few years, with standard software that can be updated regularly across the board. And that’s how Koulakov envisions Deep Nose—an electronic olfactory AI that can function as a nose that picks up scents and as a brain that interprets them. That, of course, is no easy feat. Deep Nose is modeled after the human brain, but scientists have yet to figure out how the human brain identifies one scent from another.
Smell biology
Biologically, the act of smelling is more complex and less understood than our ability to see. Recognizing a scent is a precise and intricate process in which chemistry, biology, and physics must play together in a synchronized concerto—whether you’re relishing the aroma of a rose or pinching your nose at a pile of dog poop.
Inside your nasal cavity, millions of olfactory neurons are waiting for the next smelly molecule to fly in. These neurons have microscopic finger-like protrusions called cilia, which float in the mucus covering the surface of the nasal cavity. The neurons’ other ends, called axons, stretch upward, passing through unique passages inside the skull all the way to the brain, leading to the region called the olfactory bulb (named so for its onion-like shape). When molecules fly into our nose, they bind to the cilia, and the neurons send this information to the olfactory bulb, which interprets it, resulting in our sensation of the smell. It would also pass these signals to the olfactory cortex, which would determine the smells’ quality and concentration.
Recognizing a scent is a precise and intricate process in which chemistry, biology, and physics must play together in a synchronized concerto.”
Some odor molecules bind to certain receptors but not to others. Depending on the specific combination of receptors the molecules lock onto, we would smell roses or dog poop. But even that seemingly simple molecular handshake remains mysterious. Some scientists believe in the “steric binding theory,” which states that the molecules fit receptors’ distinct physical shapes. Others support the “vibrational theory” which purports that olfactory receptors detect the molecules’ vibrational frequency and “translate” them into odors. “The steric theory suggests that there is a binding pocket of a particular shape, and some molecules will fit there, while others may swim away in the mucus,” Koulakov says. The artificial nose will require some sort of chemical sensors to detect odorant and send electrical signals to its electronic brain: the Deep Nose network that will interpret what molecules have been detected.
Fluorescent smells
Regardless of which receptor theory proves correct and whatever form artificial detectors take, Deep Nose builders face another huge challenge: designing an artificial odorant interpreting brain. Koulakov envisions it functioning as a network of multiple layers that will recognize different parts of the molecules and different chemical groups within them—just like different neurons react to the presence of different molecules inside biological brains.
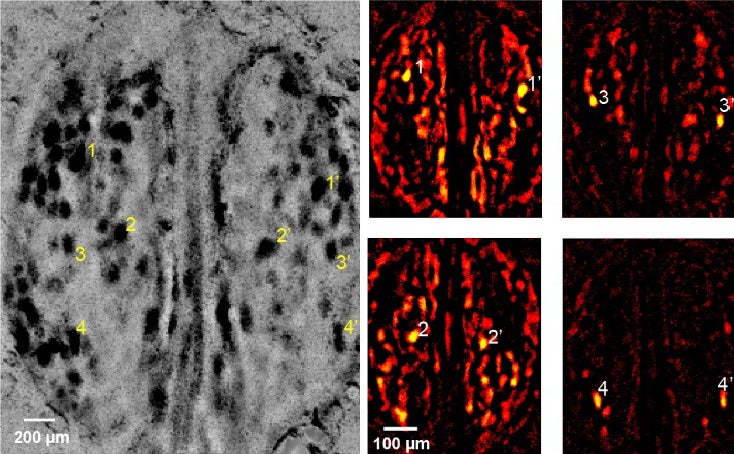
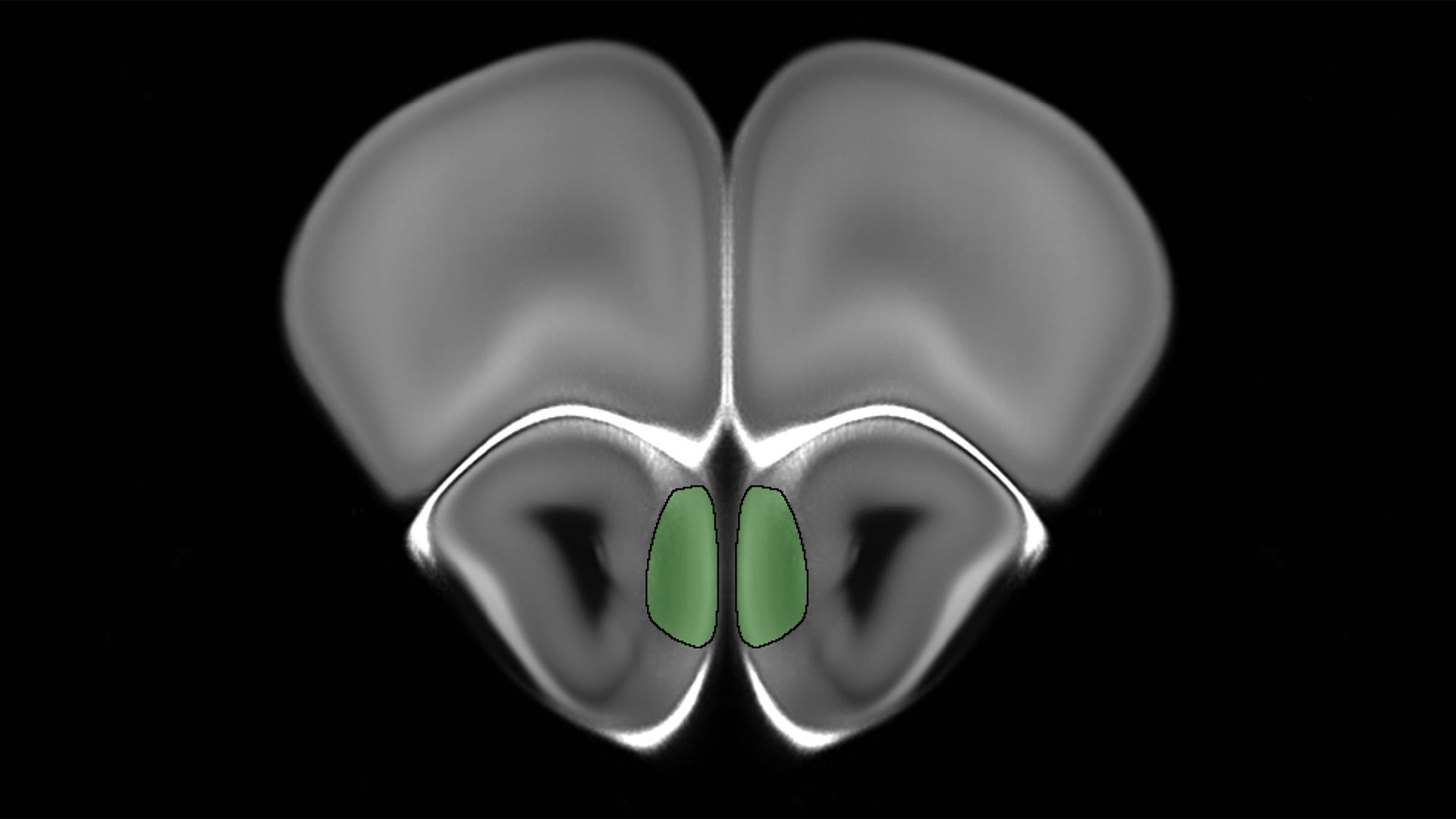
Systematically gathering neuron activation patterns helps scientists catalog the olfactory response to everything from roses to poop and from coffee to the wet-dog smell—and all other things in the “smelliverse.” Similarly, specific neuron combinations would also light up in response to particular metabolites we produce in health and disease.
Our evolution may not have designed us to diagnose disease, but we can design a software that can do so.”
Alexei Koulakov, Ph.D.
Koulakov thinks diseases will likely emit a variety of molecules. So here, rodents’ abilities would be particularly helpful. Their superb olfactory receptors that outnumber ours three-fold would let them smell many more mixtures than we can. So they can help train Deep Nose on various smells that we emit but can’t detect on our own. Just like rats have been trained to detect tuberculosis, they can be trained to sniff tumors. Researchers can map the neurons that light up in their brain in response to different cancers’ smells. “Once we collect the info about what neurons activate in response to what smells in mouse brains, we can train Deep Nose on that data,” Koulakov says. “It is important to map this ‘olfactome.’”
Science is still decades away from electronic olfactory diagnostics. However, a small army of rodents with neurons that glow in response to certain smells could help detect health ailments in about ten years, Koulakov estimates. That’s because the technology needed for observing their colorful neuronal responses already exists, but the technology necessary for mimicking the chemical sensors in the nose is yet to be created. But once this is accomplished, building an electronic nose to sniff out health problems would be fairly straightforward. “Our evolution may not have designed us to diagnose disease,” Koulakov says, “but we can design a software that can do so.”
Written by: Lina Zeldovich, Science Writer | publicaffairs@cshl.edu | 516-367-8455