Fragments snipped from tRNAs protect embryonic stem cells while they’re being epigenetically reprogrammed
Cold Spring Harbor, NY — Our genomes are minefields, studded with potentially damaging DNA sequences over which hundreds of thousands of sentries stand guard. These sentries, called epigenetic marks, attach to the double helix at such spots and prevent the underlying DNA sequences from springing into destructive action.
About half the human genome is composed of these damaging sequences. They are places where ancient viruses and parasitic elements called transposons and retrotransposons have incorporated themselves over the long course of evolution. It’s astonishing, then, to consider that during two of the most crucial processes in the life cycle, the sentries are removed, leaving the genome naked. The defenders are quickly welcomed back, but only after an interval in which the epigenetic slate is wiped clean.
Today in Cell, a team from Cold Spring Harbor Laboratory (CSHL) describes its discovery of what might be considered emergency replacements for the sentries, shock troops pressed into service across the genome only during these curiously undefended moments. Specifically, these defenders are protecting the genome in mammalian embryos, at the very early point in their development before they are implanted in the wall of the maternal uterus.
The preimplantation embryo is one of two normal settings in which epigenetic marks are wiped clean before being reinscribed. The other setting is a step in the formation of germline cells—sperm and eggs—which have temporary defenders already known to biology, so-called piwi-interacting RNAs (piRNAs). The research published today, led by first author Andrea Schorn, a postdoctoral researcher in the lab of Rob Martienssen, demonstrates that another species of small RNA performs an analogous genome-defending role in preimplantation embryos during an interval of epigenetic reprogramming. Dr. Martienssen is a CSHL Professor and Howard Hughes Medical Institute investigator.
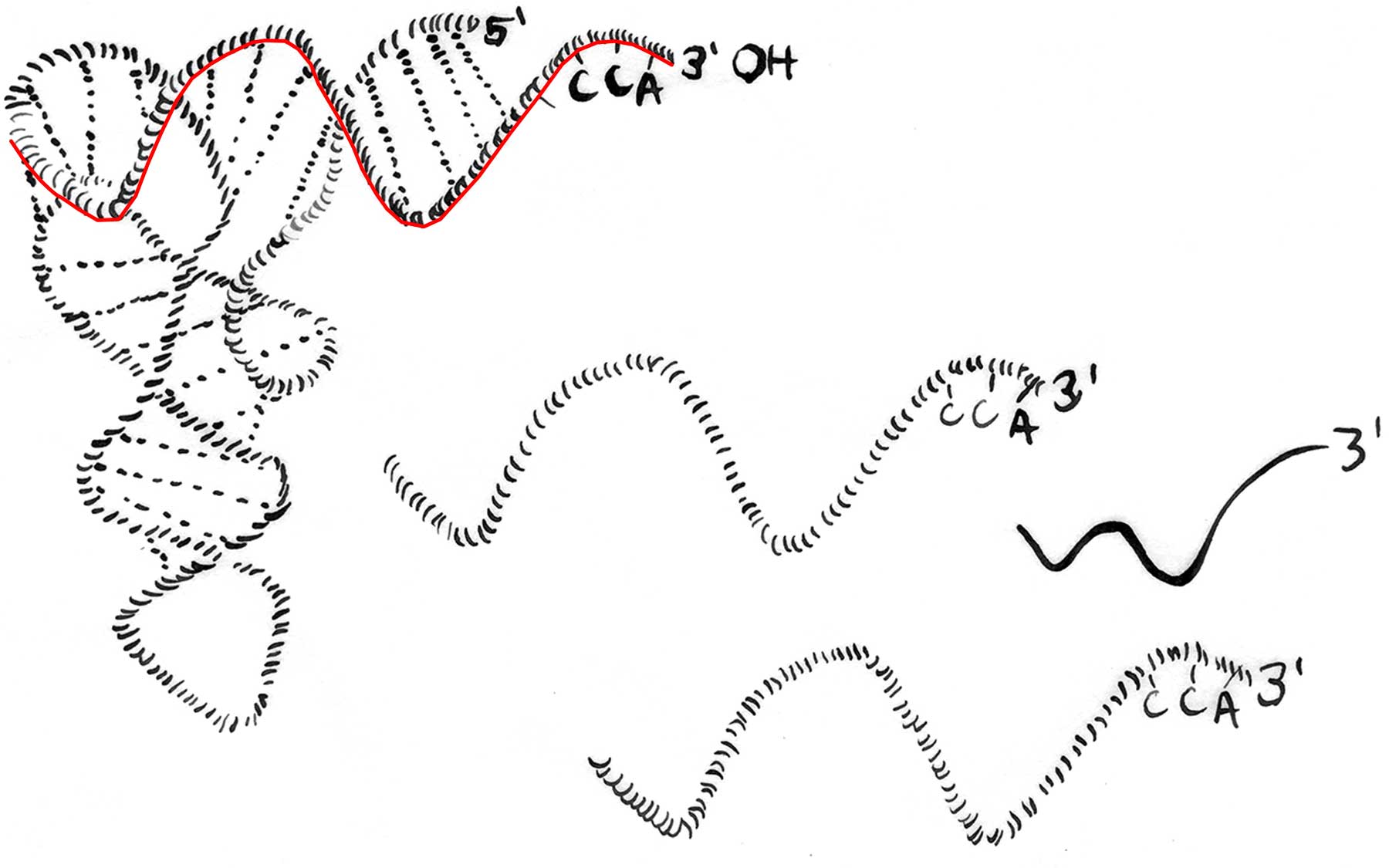
The newly identified defenders come in two varieties—RNA fragments consisting of 18 and 22 nucleotides. These RNA fragments, Dr. Schorn discovered, are perfect complements of sequences in retrotransposons that must be engaged in order for the genomic parasites to be activated.
This fact led to the discovery. Schorn scrutinized the contents of mouse embryonic stem cells and found many free-floating RNA fragments 18 nucleotides in length. Computer analysis revealed that their sequences perfectly matched sequences within transfer RNAs. tRNAs are ubiquitous, and are involved in the synthesis of proteins. It has been known for decades that tRNAs are hijacked by long terminal repeat (LTR)-retrotransposons, a portion of their sequence docking at a primer binding site (PBS) and initiating a process that activates the genomic parasite.
“Knowing that LTR retrotransposons need tRNAs to replicate, it was very tempting to believe that these 18-nucleotide tRNA fragments we were seeing in preimplantation embryonic stem cells could interfere with that process,” says Schorn. “We think the cell is deliberately chopping up full-length tRNAs into smaller fragments precisely because both tRNAs and the fragments cut from them recognize the PBS. This means the small, tRNA-derived fragments would be able to occupy that site and inhibit retrotransposon replication and mobility,” Martienssen explains.
The implications, Martienssen says, are potentially profound. This appears to tell us one way in which the genomes of mammals have tolerated vast numbers of transposons and other parasitic elements, even during periods when the genome is wiped clean of repressive epigenetic marks. “It’s plausible that this is a very ancient mechanism that cells have found to not only inhibit retrotransposons but help in protection against viruses as well,” Martienssen says.
Written by: Peter Tarr, Senior Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
The research discussed here was supported by NIH grant R01GM076396; a Bristol-Myers Squibb fellowship from the Watson School of Biological Sciences; the Howard Hughes Medical Institute; the Gordon and Betty Moore Foundation (GMBF3033); and CSHL Cancer Center Support Grant (5PP30CA045508).
Citation
“LTR-Retrotransposon Control by tRNA-Derived Small RNAs” appears online in Cell June 29, 2017. The authors are: Andrea J. Schorn, Michael J. Gutbrod, Chantal LeBlanc and Rob Martienssen. The paper can be accessed at: http://www.cell.com/cell/newarticles
Principal Investigator

Rob Martienssen
Professor & HHMI Investigator
William J. Matheson Professor
Cancer Center Member
Ph.D., Cambridge University, 1986