Cold Spring Harbor, NY — Imagine if every time you got in your car, you fired it up, put it in drive, slammed on the gas, and didn’t let up until you reached your destination. Now imagine every driver on the road did the same thing. It would be pile up after pile up.
A similar kind of chaos would reign in the brain if there were no mechanisms for slowing down its electrical signals at precisely the right time. And just like on the road, where there are multiple ways to reduce a car’s speed—stop signs, traffic lights, speed bumps—the brain has a multitude of inhibitory neurons whose job is to place checks on the brain’s excitatory neural signals.
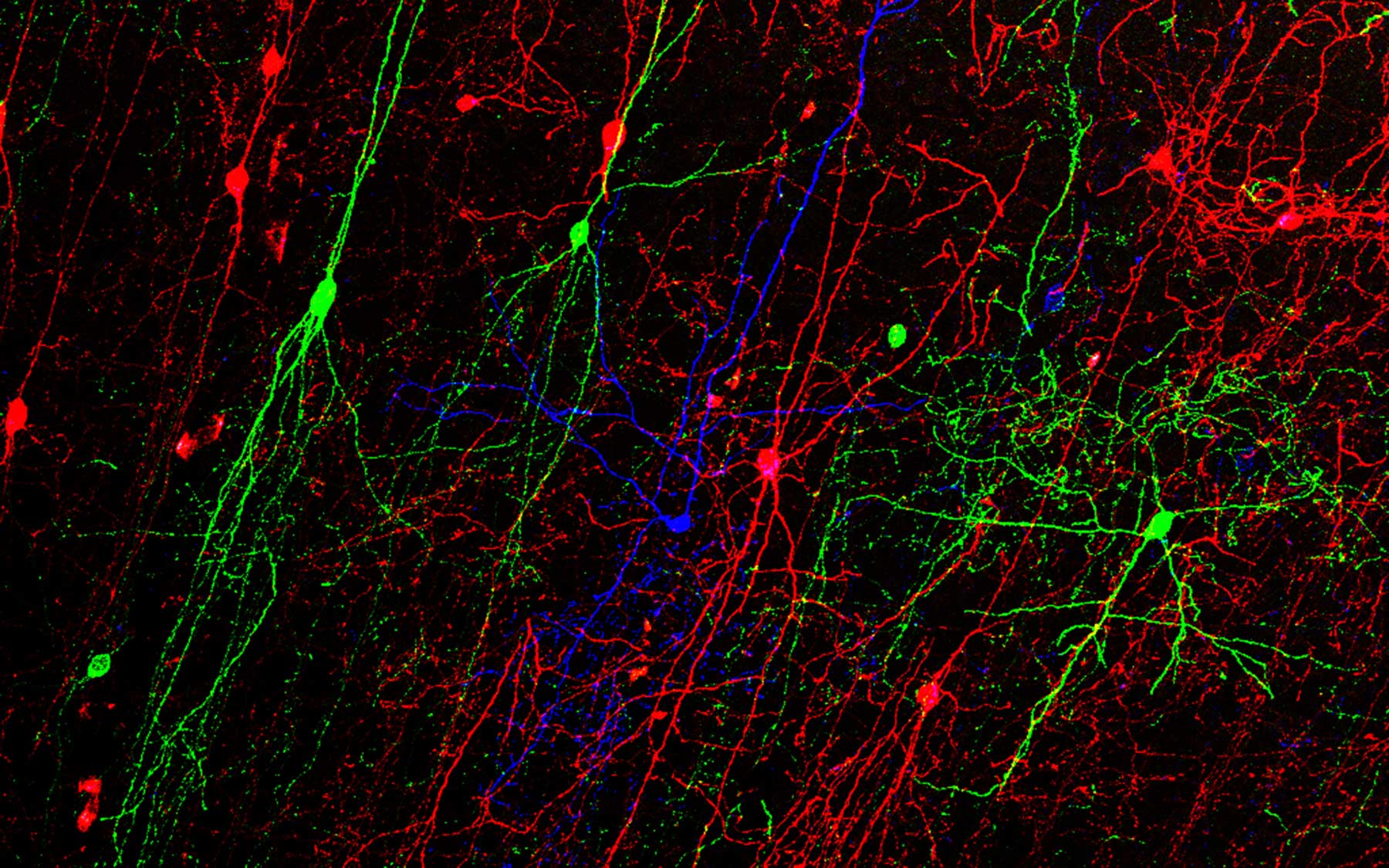
The brain’s primary inhibitory neurons—dubbed GABA neurons after the primary chemical that carries their messages—are among the most studied neurons in the brain. However, scientists do not yet have the tools necessary to monitor or modify the activity of each GABA neuron type. This level of control is thought to be necessary to understand how these neurons contribute to neurological disorders as well as the normal function of the brain. Now, researchers at Cold Spring Harbor Laboratory (CSHL) have created an innovative set of tools that combine a small handful of genetic markers and virus particles to target GABA cell types with a high degree of specificity.
“In order to understand a complex system, whether it’s an electronic device or computer, you first need to know the parts: the capacitor, resistor, the transformer,” says CSHL Professor Josh Huang, who led the team that developed the new tools. “You have to have those before you get any real understanding of how the system works.”
Most previous attempts to come up with a parts list of GABA neurons have focused on differences in the cells’ shape, projection patterns, and physiological properties. In the past decade, researchers have also begun developing tools that sort neurons according to the genes they express. However, until now such tools have lacked sufficient specificity.
“The problem is that there’s no simple correlation between one gene—or one molecular marker—and one cell type,” says Huang. “It’s more likely that multiple genes define or demarcate each cell type.”
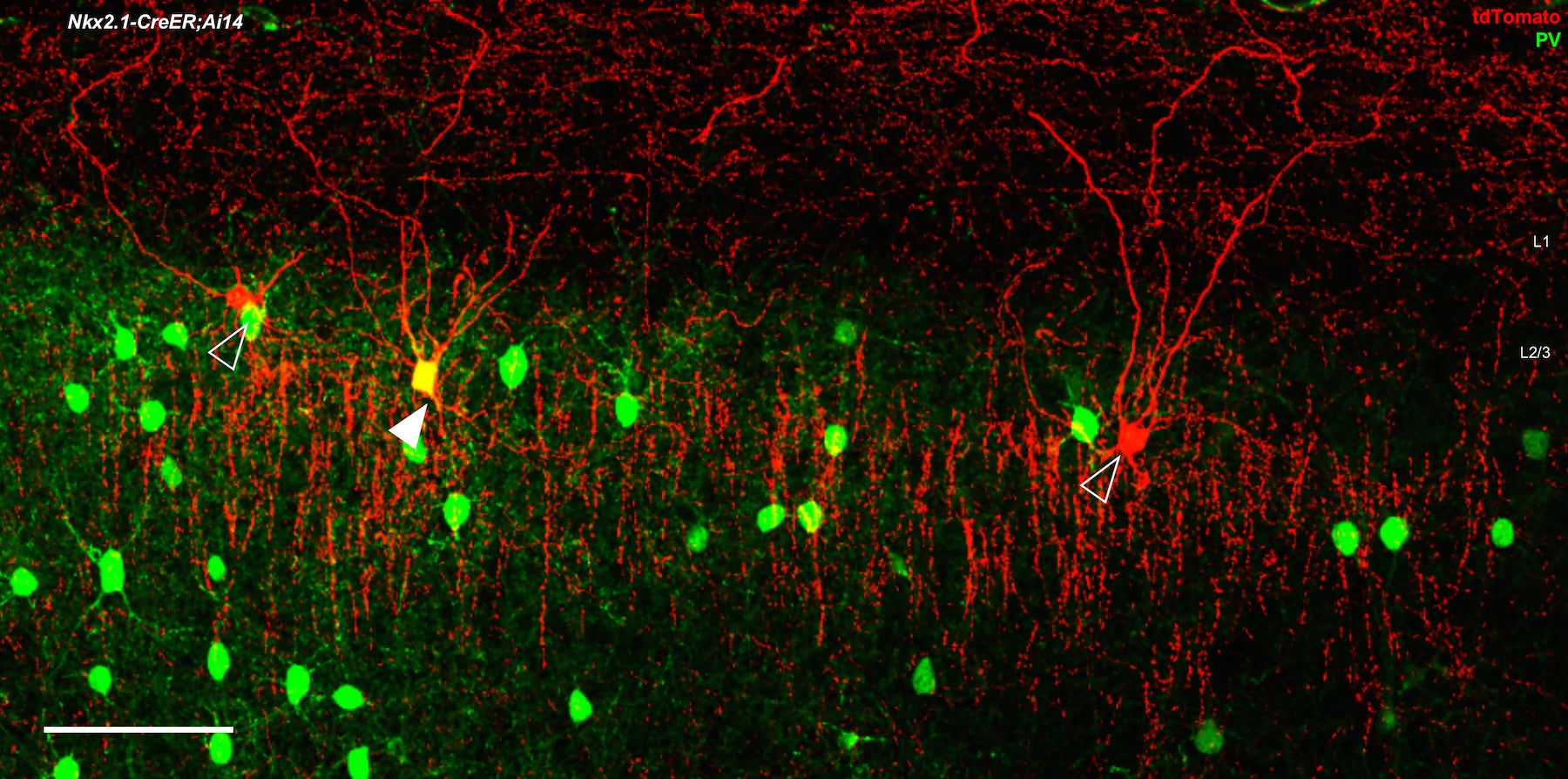
Unfortunately, practical limitations in genetic engineering have meant that researchers cannot simply increase the number of genes they search for in order to increase cell-type specificity. Therefore, Huang and his team decided they had to combine a technique for targeting just a couple of genes with a more innovative targeting method.
The key was realizing that defining a cell type requires more than just knowing its molecular characteristics. To also get at other defining characteristics, the researchers experimented with introducing various virus particles to GABA neurons that select for certain anatomical features as well as certain cell lineages and birth times during early brain development. Using this combinatorial approach, the team created a set of tools, described today in Neuron, that can identify a number of specific GABA cells by searching for just two to three genes and introducing the appropriate targeting virus.
Huang and his colleagues are now using their new set of tools to investigate the role of a specific class of GABA neurons, called chandelier neurons, in disorders such as epilepsy and schizophrenia. In addition, genetically engineered mice incorporating the novel tools are being distributed to hundreds of labs around the world. Huang thinks it might be possible to use similar tools in other species, including rats and primates.
Huang’s team is already working on another collection of tools that will systematically target different types of excitatory glutamatergic neurons, which Huang says are possibly more diverse and less understood than GABA neurons.
Written by: Chris Palmer, Science Writer | pubaff@cshl.edu | 516.367.8455
Funding
This work was supported by the National Institutes of Health, the Cold Spring Harbor Laboratory Robertson Neuroscience Fund, the National Natural Science Foundation of China, and a NARSAD Young Investigator Grant.
Citation
“Strategies and tools for combinatorial targeting of GABAergic neurons in mouse cerebral cortex,” appears online in Neuron on September 21, 2016. The authors are: Miao He, Jason Tucciarone, SooHyun Lee, Maximiliano José Nigro, Yongsoo Kim, Jesse Maurica Levine, Sean Michael Kelly, Illya Krugikov, Priscilla Wu, Yang Chen, Lin Gong, Yongjia Hou, Pavel Osten, Bernardo Rudy, and Z. Josh Huang. The paper can be accessed at: http://www.cell.com/neuron/home