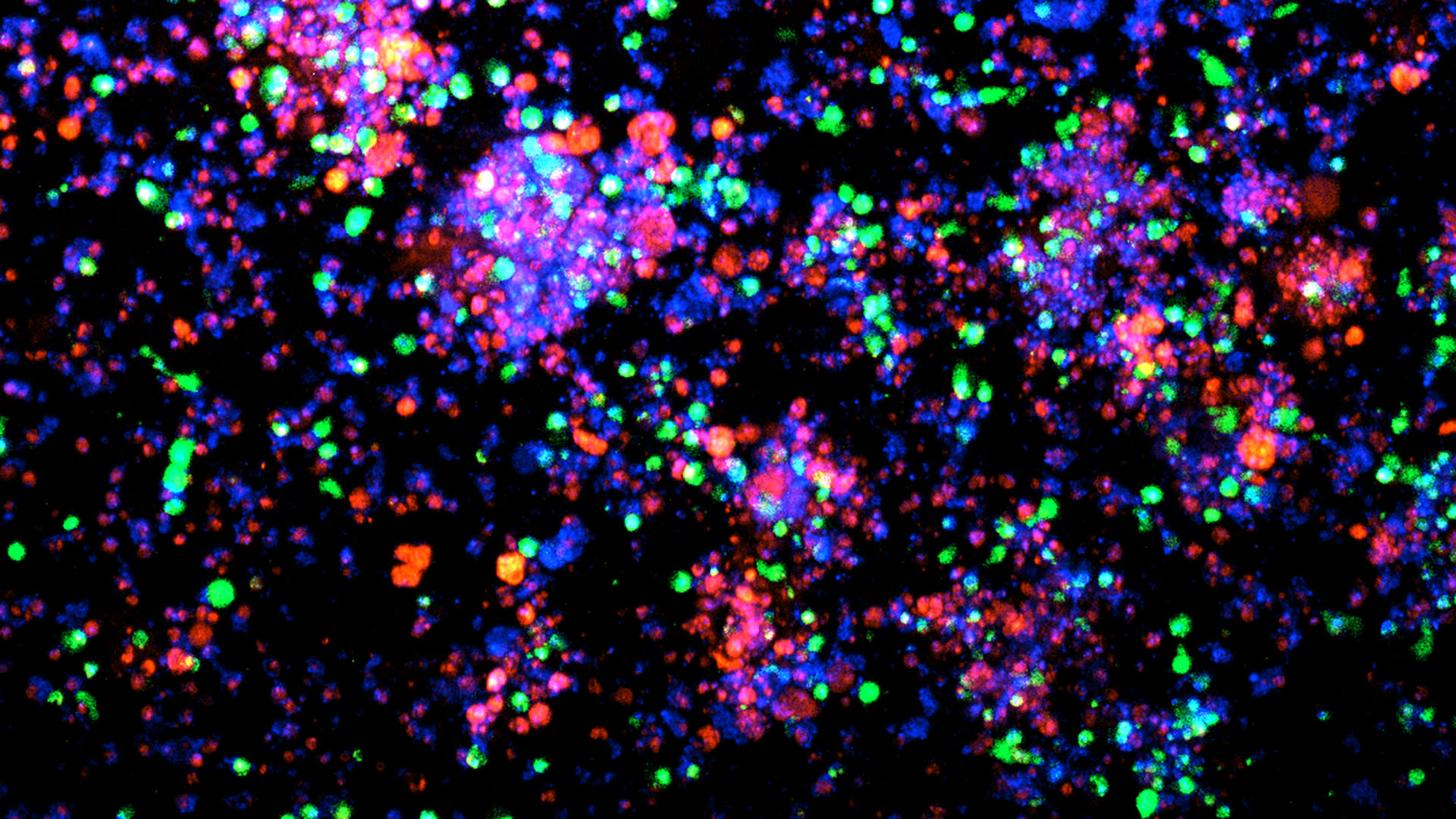
More than 68,000 women will be diagnosed with endometrial cancer in 2026. More than 14,000 will die from this condition. While some forms of the disease are treatable, high grade endometrial cancer often resists available immunotherapies.
“High grade endometrial cancers (HGEC) are very aggressive,” explains Cold Spring Harbor Laboratory Associate Professor Semir Beyaz. “Even when they respond to treatment, they often come back and have a very high mortality rate—especially in women of African ancestry. But to develop more effective therapeutics, we need better model systems to study the disease in depth.”
To close the gap, Beyaz and his team built a first-of-its-kind research platform containing matching pairs of endometrial cancer organoids and immune cells. They collaborated with Northwell Health physicians Gary Goldberg and Marina Frimer to obtain patient specimens. Eighty-five volunteers provided tissue and tumor samples along with corresponding immune cells. Using the new platform, Beyaz and his team discovered that HGEC evades the immune system by blocking a protein called MHC II.
“MHC is part of an immune surveillance mechanism that is crucial for detecting cancer, almost like a passport or identity card,” Beyaz says. “HGEC cells dampen MHC II molecules specifically. When they hide that molecule, immune cells cannot recognize endometrial cancer. So, we said, ‘OK, how can we restore that?’”
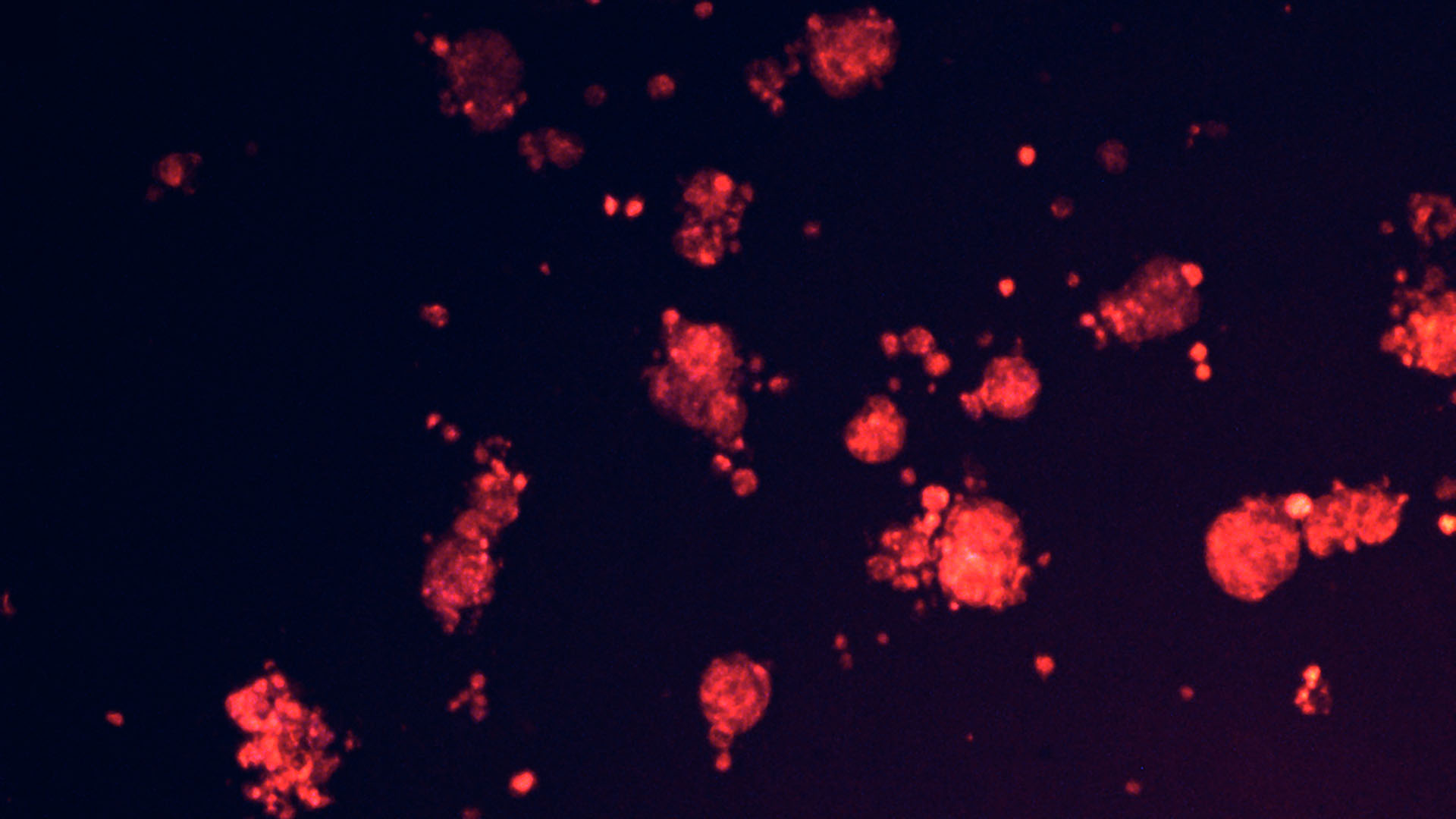
In one series of experiments, the Beyaz lab used a drug called Tazemetostat, which the FDA had approved for use against certain blood cancers, to help to turn on genes that are silent in cancer cells. Reactivating MHC II kickstarted the immune response against endometrial cancer. Notably, Tazemetostat was recently withdrawn over safety concerns. Nevertheless, these results are promising.
“Finding already FDA approved drugs have the potential to improve efficacy of immunotherapy in endometrial cancer is very exciting!” Beyaz says.
Because the new platform includes matched tumor organoids and immune cells, it allows researchers to test the safety and efficacy of emerging treatments. Beyaz and his team next utilized CAR T cells recognizing a molecule called EGFR that is highly abundant in HGEC cells but less so in normal endometrial cells. These CAR T cells robustly killed the HGEC cells but spared the normal cells.
“That’s the beauty of our system,” Beyaz says. “You can take any drug of interest and test it on the disease as it exists in patients across the population.”
The Beyaz lab’s new platform lays the foundation for scientists and the biotech industry to expand research efforts into HGEC. They’ve already seen early success. In collaboration with Revitope Oncology, Beyaz’s lab tested a next generation immunotherapy modality called protease-activatable bispecific T-cell engagers (TCEs) against HGEC organoids.
Beyaz now hopes to extend this platform to other, more common women’s health conditions, including endometriosis. “Endometrial cancers and endometriosis both suffer from a lack of investment and research,” he says. “Our ultimate goal is to bring more awareness and resources to the fight against these painful and devastating diseases. This new platform opens the door.”
Written by: Nick Wurm, Communications Specialist | wurm@cshl.edu | 516-367-5940
Funding
National Cancer Institute, Oliver S. and Jennie R. Donaldson Charitable Trust, Mark Foundation for Cancer Research, CSHL-Northwell Health Affiliation, New York Genome Center Polyethnic-1000 Initiative, Revitope Oncology
Citation
Chung, C., et al., “Ancestrally diverse autologous patient-derived organoid – immune cell co-culture platform for addressing immunotherapeutic outcome disparities in high-grade endometrial cancer”, Cancer Research Communications, February 19, 2026. DOI: 10.1158/2767-9764.CRC-25-0193
Core Facilites
Principal Investigator

Semir Beyaz
Assistant Professor
Cancer Center Member
Ph.D., Harvard University, 2017