Cold Spring Harbor, NY — Scientists at Cold Spring Harbor Laboratory (CSHL) today report a discovery that they hope will lead to the development of a powerful new way of treating an aggressive form of breast cancer.
The breast cancer subtype in question is commonly called “HER2-positive”; it’s a subset of the disease affecting about one patient in four, in which tumor cells overexpress a signaling protein called HER2. The blockbuster drug Herceptin is a first-line treatment for many women with HER2-positive breast cancer, but in most cases, resistance to the treatment develops within a year.The prognosis for HER2-positive breast cancer patients is worse than for those with other subtypes of the illness.
In a paper appearing online today in Nature Chemical Biology, a multi-institution team led by CSHL Professor Nicholas Tonks reports that it has found a means of inhibiting another protein, called PTP1B, whose expression is also upregulated in HER2-positive breast cancer. They show that PTP1B plays a critical role in the development of tumors in which HER2 signaling is aberrant. Therefore, PTP1B may be a therapeutic target through which to treat the disease.
When they treated mice modeling HER2-positive breast cancer with a drug candidate called MSI-1436 (also called trodusquemine), Tonks and colleagues not only inhibited signaling by PTP1B, but also signaling by HER2 proteins.
“The result was an extensive inhibition of tumor growth and prevention of metastasis to the lung in HER2-positive animal models of breast cancer,” notes Navasona Krishnan, Ph.D., a postdoctoral investigator in the Tonks lab who performed many of the experiments and is lead author on the paper reporting the results.
Dr. Tonks discovered PTP1B some 25 years ago. It is an enzyme—one in a “superfamily” of 105 called protein tyrosine phosphatases (PTPs)—that perform the essential biochemical task of removing phosphate groups from amino acids called tyrosines in other proteins. Adding and removing phosphates is one of the principal means by which signals are sent among proteins.
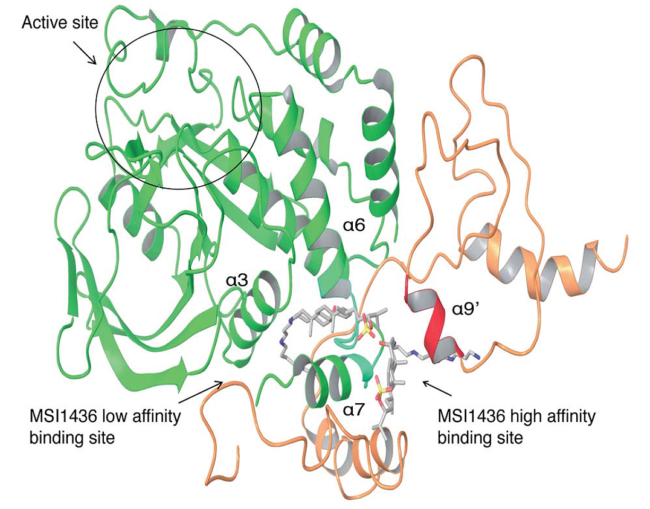
PTP1B for many years has been a target of interest among drug developers. It is well known as a so-called negative regulator of insulin action—an antagonist of insulin signaling, as well as signaling by leptin, the hormone that helps regulate appetite. Drugs that can block or inhibit the action of PTP1B have enormous potential in controlling diabetes and obesity. Yet properties of the molecule—in particular involving the charge of its active site—have stymied potential developers of inhibitors.
The new paper by Tonks and collaborators importantly reveals an alternative binding site, called an allosteric site, that does not present the biochemical difficulties that the main, or “catalytic,” binding site does. It is this allosteric site that is the target of the candidate drug trodusquemine.
Later this year early-stage human trials will begin for the drug, a collaboration of CSHL and clinicians at the Monter Cancer Center of North Shore-Long Island Jewish Hospital. Dr. Tonks and CSHL have interests in a joint venture called DepYmed Inc., in partnership with Ohr Pharmaceutical (NasdaqCM: OHRP). The venture seeks to develop trodusquemine and related analogs.
Written by: Peter Tarr, Senior Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
Funders for the research discussed in this release include: the National Institutes of Health, Cold Spring Harbor Laboratory Cancer Center, American Diabetes Association, Brown University Research Seed Fund, and Agence Nationale de Researche.
Additional funders of research in the Tonks lab include: Aboff’s Inc., F.A.C.T. Long Island Foundation, Glen Cove C.A.R.E.S., Hearts for Cancer, Islip Breast Cancer Coalition, The Irving A. Hansen Memorial Foundation, Jo-Ellen and Ira Hazan, Joni Gladowsky Breast Cancer Foundation, The Don Monti Memorial Research Foundation, Masthead Cove Yacht Club in Memory of Carol Marcincuk, Panera Breads, Fannie E. Rippel Foundation, Judi Shesh Memorial Fund, Constance Silveri, Mary F. Smith Family Foundation & West Islip Breast Cancer Coalition.
Citation
“Targeting the disordered C terminus of PTP1B with an allosteric inhibitor” appears May 21, 2014 in Nature Chemical Biology. The authors are: Navasona Krishnan, Dorothy Koveal, Daniel H. Miller, Bin Xue, Sai Dipikaa Akshinthala, Jaka Kragelj, Malene Ringkjobing Jensen, Carla-Maria Gauss, Rebecca Page, Martin Blackledge, Senthil K. Musthuswamy, Wolfgang Peti and Nicholas K. Tonks. the paper can be obtained at: http://www.nature.com/nchembio/journal/vaop/ncurrent/full/nchembio.1528.html
Principal Investigator

Nicholas Tonks
Professor
Caryl Boies Professor of Cancer Research
Cancer Center Associate Director of Shared Resources
Ph.D., University of Dundee, 1985
