A team of scientists led by CSHL Professor David L. Spector, Ph.D., has discovered how “bookmarking” a gene pre-division accelerates its reactivation post-division.
Cold Spring Harbor, NY — In order for cells of different types to maintain their identities even after repeated rounds of cell division, each cell must “remember” which genes were active before division and pass along that memory to its daughter cells. Cells deal with this challenge by deploying a “bookmarking” process. In the same way a sticky note marks the last-read page in a book, certain molecules tag the active genes in a cell so that, after it divides, the same genes are reactivated right away in the new cells.
“What we didn’t know, however, was how bookmarking a gene pre-division causes it to get reactivated post-division,” says CSHL Professor David L. Spector, Ph.D. By observing and measuring the kinetics of activation of a single gene locus—the specific chromosomal location of a gene—in a cell before it divides and comparing it with the same gene’s reactivation in newly formed daughter cells, Spector and his team have now arrived at the answer to this question. Their study appears online ahead of print in Nature Cell Biology on October 9.
During cell division, or mitosis, there is a temporary blackout of all gene activity within the cell. The cell’s chromatin—the coils of chromosomal DNA wrapped around histones (proteins that package DNA)—becomes tightly compacted; most proteins that normally cling to chromatin to maintain gene expression are stripped away; and transcription—the copying of DNA into RNA—comes to a halt. When division ends, the chromatin in the new cells de-compacts or relaxes, transcription-regulating proteins are recruited to specific sites in the chromatin and gene transcription begins anew.
To find out how this post-division process is set in motion, Spector’s team used an innovative live cell imaging approach that they previously developed to make real-time observations of a specific gene locus and the RNA transcribed from the locus when it is activated. In this experimental system, the gene locus under observation, the transcription-activating protein, and the RNA produced by the gene are each labeled with a different colored fluorescent tag.
“By looking at gene expression at the same locus (a gene’s position on the chromosome) at two different times—during ‘interphase,’ which is the stage between cell division, and after mitosis, we discovered that the gene is activated much more rapidly when it is reactivated post-mitosis,” explains Spector. Further analysis suggested that the transcriptional activity during interphase leaves behind a memory in the form of a bookmark that is preserved during mitosis and is crucial for the rapid post-mitotic reactivation of the gene.
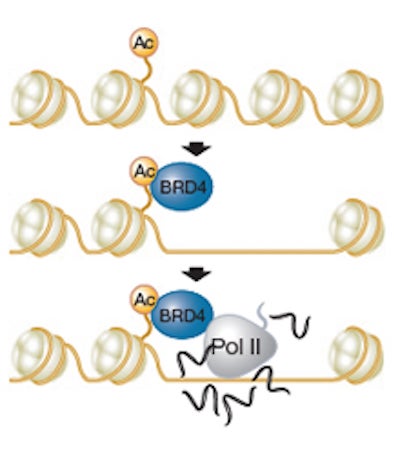
This bookmark, the scientists show, is a histone molecule that has undergone a chemical modification called acetylation, which alters its interactions with DNA as well as with proteins that bind to it. “We observed that the gene locus accumulates this acetylated histone, called H4K5Ac, prior to mitosis. During mitosis, H4K5Ac stays in place along with a small amount of another protein called BRD4, which keeps the locus less tightly packed than the rest of the chromatin,” explains the paper’s first author Rui Zhao, Ph.D.
When mitosis ends, the scientists observed additional BRD4 being rapidly recruited to the bookmarked loci in the daughter cells. This in turn loosens up the tightly wound chromatin and recruits other members of the transcription machinery to the bookmarked gene, which is then rapidly reactivated.
Previous studies have shown BRD4 to be a “reader” of chromatin marks such as acetylated histones, which might contribute to its quick buildup at the bookmarked loci, according to Spector. Another recent CSHL study identified a role for BRD4 in cancer cell proliferation, showing that inhibiting its activity stopped leukemia progression.
“Our study points to a new role for BRD4 in chromatin decompaction,” says Spector. “The fact that BRD4 recruitment precedes the arrival of RNA polymerase II at the bookmarked loci in post-mitotic cells suggests a more global role for this protein in gene reactivation in newly formed cells,” he adds. “It’s clear that the ‘early-to-activate’ genes are the ones that need to be bookmarked and there will be many different bookmarks. We are now trying to identify these genes and determine what regulates their selection for early reactivation.”
Written by: Hema Bashyam, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
This work was supported by a grant from the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health (NIH).
Citation
“Gene bookmarking accelerates the kinetics of post-mitotic transcriptional reactivation” appears in Nature Cell Biology online ahead of print on October 9. The full citation is: Rui Zhao, Tetsuya Nakamura, Yu Fu, Zsolt Lazar and David L. Spector. The paper can be downloaded at http://www.nature.com/ncb/index.html using the doi:10.1038/ncb2341
Principal Investigator

David L. Spector
Professor
Robert B. Gardner Jr., Professor
Cancer Center Member
Ph.D., Rutgers University, 1980