High blood pressure, inflammation, and the sensation of pain may rely in part on tiny holes on the surface of cells, called pores. Living cells react to the environment, often by allowing water and other molecules to pass through the cell’s surface membrane. Protein-based pores control this flow.
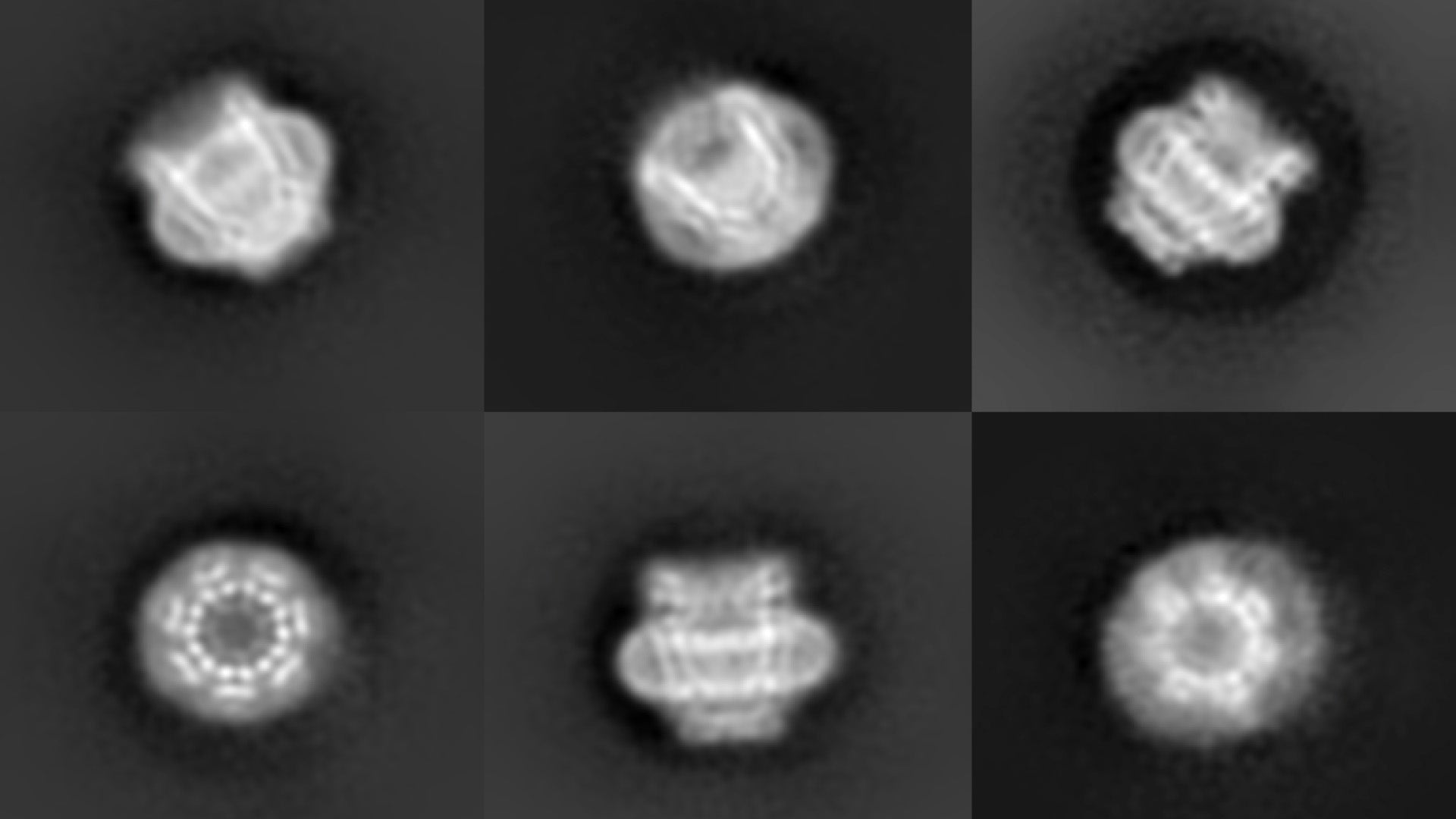
One set of pores, called pannexins, are like tiny “black holes” of biology; their existence has been confirmed by computational and functional evidence but until very recently, they were never actually seen. They help cells communicate with and react to their surroundings, but until scientists see what they look like, it is hard to know-how. Now, a team of researchers from Cold Spring Harbor Laboratory (CSHL) and Cornell University have developed very high-resolution images of these elusive structures.
“We all knew these pores had to exist… but there’s definitely been some mystique about pannexins,” said postdoctoral researcher Kevin Michalski, who co-led the project. “Without images, it’s been really difficult to figure out exactly what these pores are doing.”
Michalski spent more than six years trying to get a clear image of pannexins.
“I had spent so long trying to set up experiments without really being able to see what I was working with. That’s what makes this so exciting,” Michalski explained. “For the first time, we can definitely see this detailed structure—it’s all new information.”
Michalski and Johanna Syrjanen, also in the Furukawa lab at CSHL collaborated with the Kawate lab at Cornell University, and published their findings in the journal eLife. They used a technique called cryo-electron microscopy (cryo-EM) to capture a half-million images of a pannexin pore, called “pannexin 1,” from many different angles. They then merged the images to form a three-dimensional model. Pannexin 1 is made up of seven identical proteins in a donut-like ring around a central pore.
Researchers have hypothesized that pannexin 1 is responsible for releasing the signals that trigger a failing cell’s self-destruction. These signals prompt macrophages to destroy the ailing cell, clearing the way for new cells to thrive. Without the replacement of ailing cells with new cells, organisms age.
“But what’s interesting is that even cells that don’t go through the process of self-destruction can have pannexins,” Michalski said. “That means that there has to be some other important mechanisms that this pore is performing.”
“The way pannexin 1 is shaped on a cell is different from another kind of pore that my lab recently revealed,” added Professor Hiro Furukawa, who oversaw the research. “It appears that there are more kinds of pores than scientists originally anticipated. How and when they open and close are important questions to be explored.”
The flow of water, salts, and other molecules into a cell across a pore regulates the cell’s size. Cells that misregulate their size or water flow could contribute to high blood pressure, pain, and inflammation. Pannexins are also thought to allow much larger molecules to pass through their pores. Understanding how molecules negotiate their way through a pannexin could lead to new classes of drugs.
Written by: Brian Stallard, Content Developer/Communicator | publicaffairs@cshl.edu | 516-367-8455
Funding
This work was supported by the National Institutes of Health (NIH), the Robertson funds at Cold Spring Harbor Laboratory, the Doug Fox Alzheimer’s fund, Austin’s Purpose, and the Heartfelt Wing Alzheimer’s fund.
Citation
Michalski et al, “The cryo-EM structure of a pannexin 1 reveals unique motifs for ion selection and inhibition,” eLife, 12 Feb 2020.
Principal Investigator

Hiro Furukawa
Professor
Cancer Center Member
Ph.D., The University of Tokyo, 2001