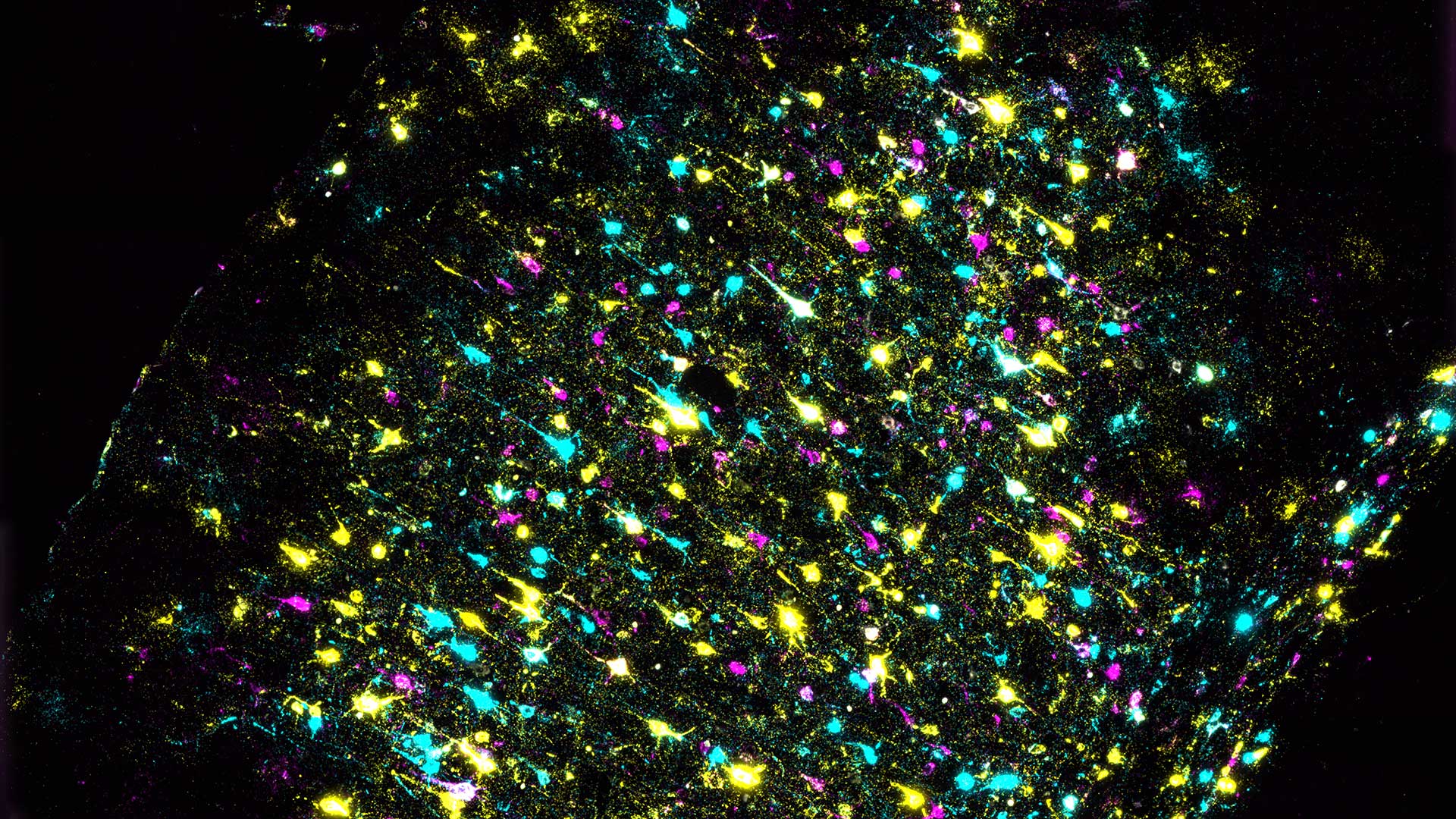
A new method developed at Cold Spring Harbor Laboratory (CSHL) uses DNA sequencing to efficiently map long-range connections between different regions of the brain. The approach dramatically reduces the cost of mapping brain-wide connections compared to traditional microscopy-based methods.
Neuroscientists need anatomical maps to understand how information flows from one region of the brain to another. “Charting the cellular connections between different parts of the brain—the connectome—can help reveal how the nervous system processes information, as well as how faulty wiring contributes to mental illness and other disorders,” says Longwen Huang, a postdoctoral researcher in CSHL Professor Anthony Zador’s lab. Creating these maps has been expensive and time-consuming, demanding massive efforts that are out of reach for most research teams.
Researchers usually follow neurons’ paths using fluorescent labels, which can highlight how individual cells branch through a tangled neural network to find and connect with their targets. But, the palette of fluorescent labels suitable for this work is limited. Researchers can inject different colored dyes into two or three parts of the brain, then trace the connections emanating from those regions. They can repeat this process, targeting new regions, to visualize additional connections. In order to generate a brain-wide map, this must be done hundreds of times, using new research animals each time.
This animation shows how mouse brain cells connect to one another—a sort of wiring diagram of the brain. Researchers in Anthony Zador’s lab at Cold Spring Harbor Laboratory have developed a technique to map many of these connections at once using a “DNA barcode”—a battery of short DNA snippets. Other techniques to determine how cells connect are far more laborious and expensive.
The method developed in the Zador lab, called brain-wide individual animal connectome sequencing (BRICseq), takes a different approach. “We no longer label brain regions and their projections using colors. We are labeling them using nucleotide sequences,” Huang says. Combining the four letters of the DNA code into short “barcodes” generates a virtually infinite number of labels that can distinguish one cell from one another, he explains. After labeling, researchers use DNA sequencing to analyze tiny segments of brain tissue, interpreting each recurring barcode as a signal of a cellular connection.
“The diversity of barcodes is really high compared to the number of colors we can use in science research. So now we can really label a huge amount of neurons and brain regions per animal, which allows us to map projections from multiple brain regions using these barcodes,” Huang says.
The research team, including former graduate student Justus Kebschull, who worked with Huang to develop the technique, report in the journal Cell that BRICseq accurately maps region-to-region connectivity in the brains of mice. They say the approach will be widely accessible and should be adaptable to other organisms.
Written by: Jennifer Michalowski, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
The National Institutes of Health, the Brain Research Foundation, the Simons Foundation, the Paul G. Allen Frontiers Group, the Boehringer Ingelheim Fonds, the Genentech Foundation, the Simons Collaboration on the Global Brain, and the Army Research Office.
Citation
Huang, L., et. al. “BRICseq Bridges Brain-wide Interregional Connectivity to Neural Activity and Gene Expression in Single Animals,” Cell, July 2, 2020. DOI: https://doi.org/10.1016/j.cell.2020.05.029
Principal Investigator

Anthony Zador
Professor
The Alle Davis and Maxine Harrison Professor of Neurosciences
M.D., Ph.D., Yale University, 1994