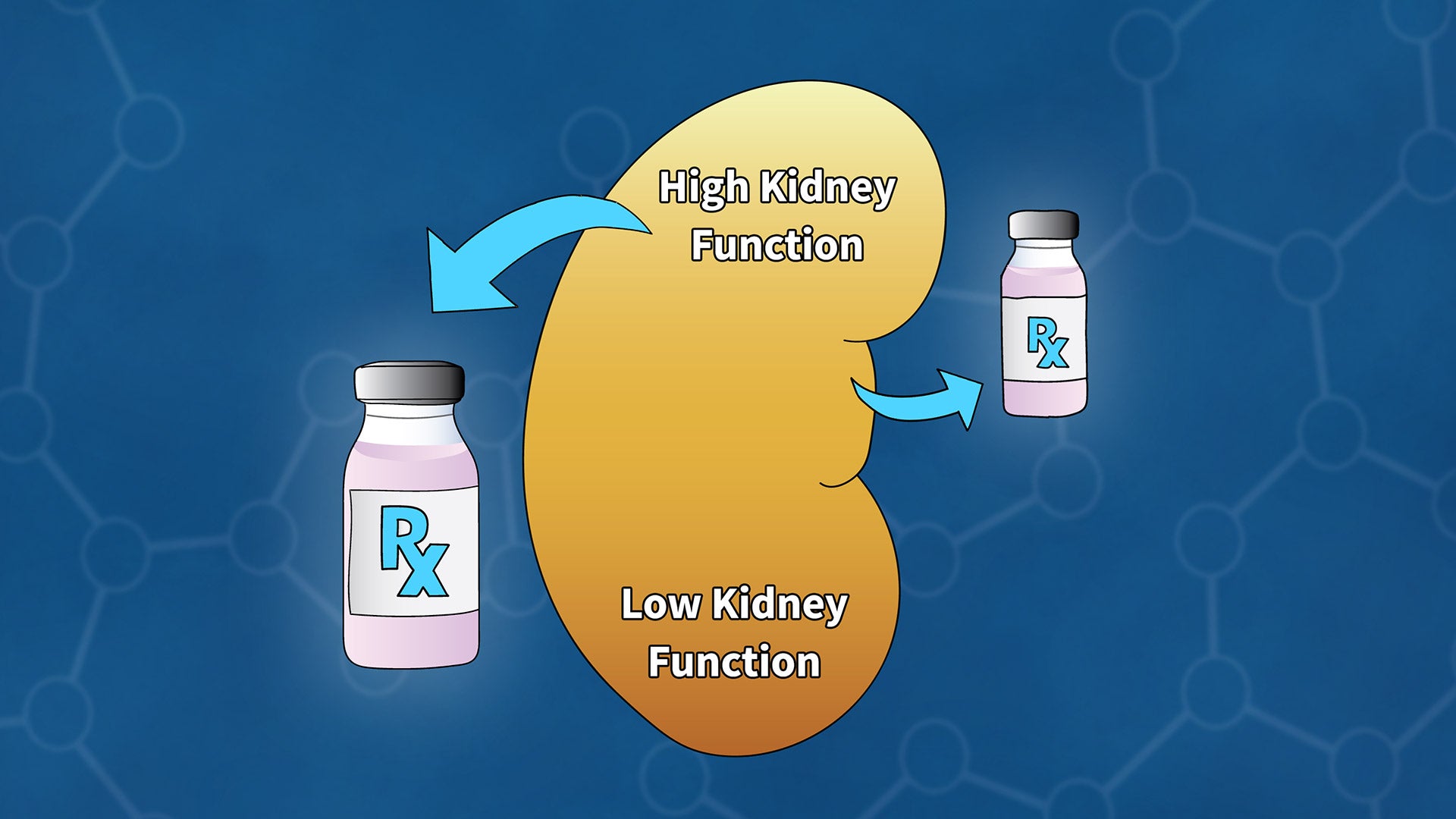
Cold Spring Harbor Laboratory (CSHL) scientists have developed a new model for assessing kidney function in cancer patients as part of an international collaboration that involved contributions from the United Kingdom and Sweden. The model gives clinicians a new tool to aid dose determinations for chemotherapy to treat a patient’s disease while limiting the drugs’ harmful effects.
Chemotherapy drugs are usually injected into the bloodstream and removed from the body as blood filters through the kidneys. Sometimes the drugs harm the kidneys. Understanding how efficiently a patient can filter blood is essential for predicting how the body will process a drug and monitoring treatment effects. To be sure a treatment reaches tumor cells at the right dose, patients whose kidneys remove compounds from their blood efficiently will need more drug than someone whose kidney function is impaired. Calculating an appropriate dose is a crucial balancing act. CSHL Assistant Professor Tobias Janowitz, the senior author of the study, says:
“This is the constant balance that we are trying to get right in cancer therapy across the board. We have to be careful to make sure that we don’t put the patient at risk. And we have to make sure that we dose the drugs in a way that we get this balance right between how effective is the drug and how many side effects does the drug cause.”
 Clinicians assess kidney function before every round of cancer treatment. Measuring the kidneys’ filtration rate directly is costly and requires specialized facilities, so instead, it is frequently estimated by measuring how much creatinine—a chemical waste product—is in the blood. The relationship between creatinine levels and filtration rate depends on several factors, including age, gender, and body mass index. Mathematical models are used to integrate these data.
Clinicians assess kidney function before every round of cancer treatment. Measuring the kidneys’ filtration rate directly is costly and requires specialized facilities, so instead, it is frequently estimated by measuring how much creatinine—a chemical waste product—is in the blood. The relationship between creatinine levels and filtration rate depends on several factors, including age, gender, and body mass index. Mathematical models are used to integrate these data.
Previous models were developed using data from patients with kidney disease, whose failing kidney function must be closely monitored. But it hasn’t been clear how those models represent what’s happening in patients with cancer, Janowitz says. “Cancer patients may have a unique physiology, and most of them don’t have impaired kidney function. So, it is conceivable that the models that are available don’t serve cancer patients so well.”
Janowitz and his colleagues, including CSHL fellow Hannah Meyer, developed their model using data from patients at three cancer centers. The new model, described in the journal Clinical Cancer Research, is compatible with two widely used methods for measuring creatinine and provides enough precision to guide clinical decisions. Follow-up studies will be needed to determine whether using the new model improves treatment outcomes for cancer patients.
Written by: Jennifer Michalowski, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
Cancer Research UK, Experimental Medicine Initiative Clinical Lectureship, National Institute for Health Research, Cold Spring Harbor Laboratory, Pershing Square Foundation, and the National Institutes of Health USA Cancer Center.
Citation
Williams, E.H., et al., “CamGFR v2: a new model for estimating the glomerular filtration rate from standardized or non-standardized creatinine in patients with cancer”, Clinical Cancer Research, December 10, 2020. DOI: 10.1158/1078-0432.CCR-20-3201
Principal Investigator

Tobias Janowitz
Associate Professor
Cancer Center Program Co-Leader
M.D., Ph.D., University of Cambridge, UK, 2007