Women consistently report that nursing is easier after a first pregnancy. Some remarkable genomic work led by Professor Greg Hannon provides a biological reason for this effect and shows more broadly how bodily experiences can prepare us to respond to future stimuli.
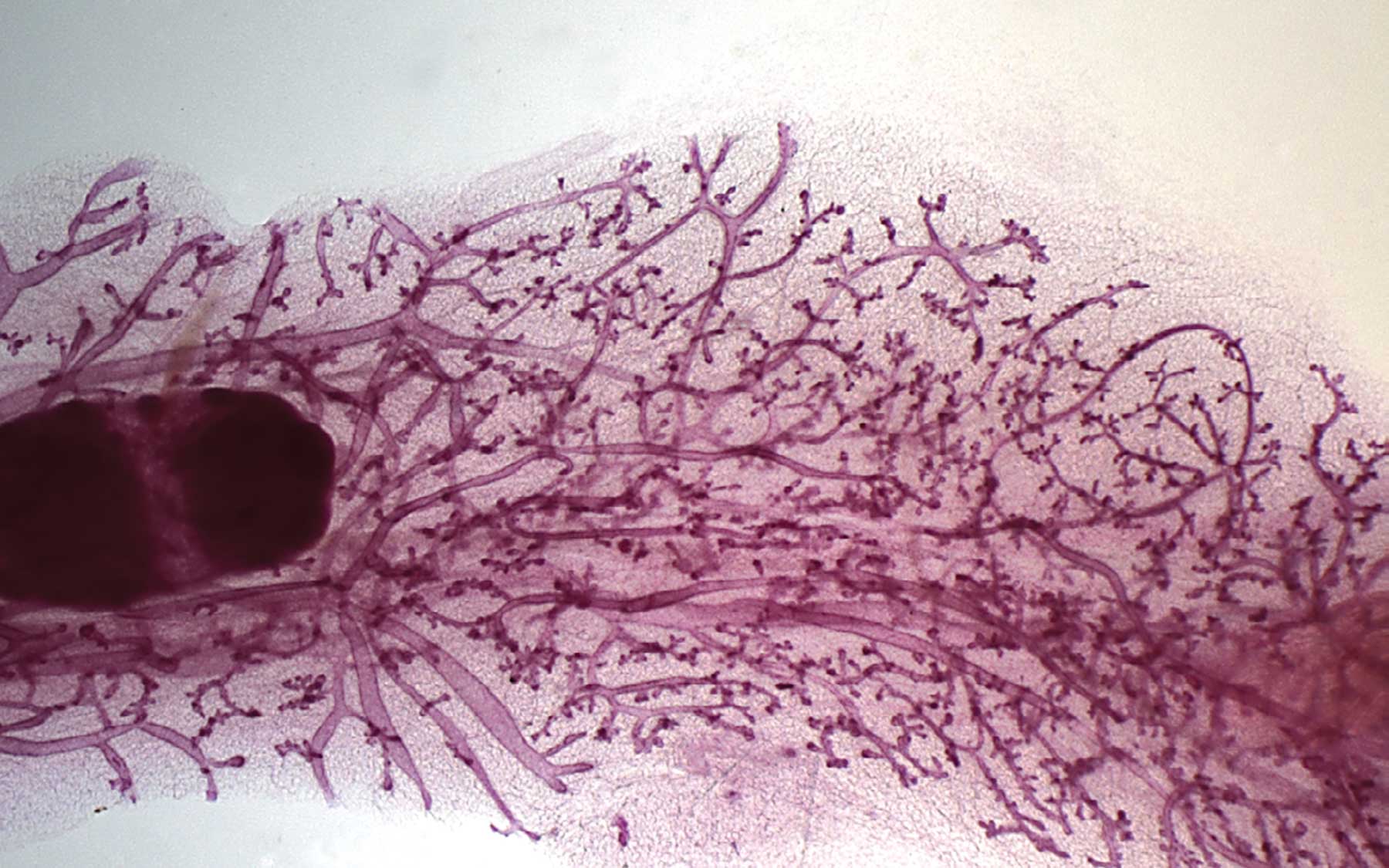
The razor-thin slice of breast tissue pictured here is sampled from a mouse 6 days after becoming pregnant. Delicate branching structures support the ducts through which milk will flow. Spurred by estrogen and progesterone, breast cells proliferate and the ductal network becomes more profuse. After nursing, the tissue “involutes”—returns to its less dense pre-pregnancy state. Yet in subsequent pregnancies, breast tissue expands much more rapidly at the first hormonal signal, and becomes so densely packed with ducts that a light shining from below can barely pass through a slice like the one shown here.
“It’s as if the gland already knows these hormones,” says Camila dos Santos, now a CSHL Assistant Professor. As a Hannon lab postdoc, dos Santos devised a technique that helped the team reveal the secret to what is in fact a life-long biological memory of an earlier pregnancy. In one experiment, dos Santos used a marker she’d discovered to classify six types of breast cells. She then prepared maps revealing all the positions along the genome where methyl molecules “marked” the double helix in each cell type, before and after a first pregnancy.
The team’s discovery: during a first pregnancy, these epigenetic marks are wiped away from many genomic positions. Those positions become occupied by a gene regulating transcription factor called Stat5a. Should hormones signal another pregnancy, breast cells already know to activate adjacent genes—which regulate cell proliferation, ductal growth and lactation. After a first pregnancy, therefore, breast cells are primed for nursing. Dos Santos now seeks to unravel the mystery of how pregnancy-induced changes, perhaps including these, protect some women from breast cancer.
Written by: Peter Tarr, Senior Science Writer | publicaffairs@cshl.edu | 516-367-8455