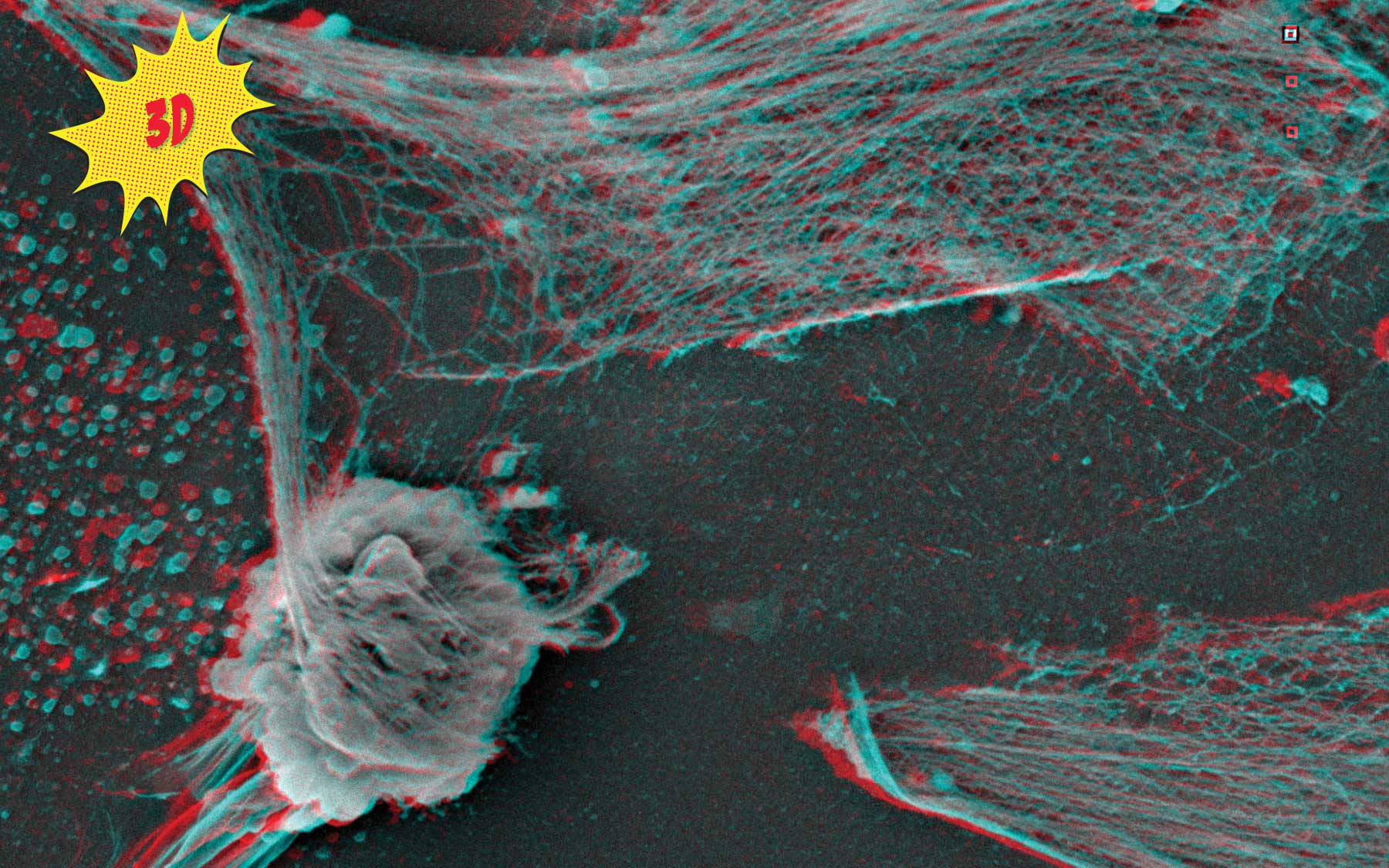
Cancer is infamous for repurposing molecules and mechanisms our body routinely uses to sustain itself. This remarkable 3D image, made by CSHL’s expert electron microscopist Stephen Hearn, dramatically captures one such mechanism, spiderweb-like structures made of DNA and studded with toxic enzymes. Normally, these webs are cast out into extracellular space by white blood cells called neutrophils—like the one in the lower left of this image. The webs snag and digest invaders such as bacteria and viruses.
Using a separate visualization technique called intravital imaging that she developed, Associate Professor Mikala Egeblad recently discovered that the lattice-like structures—appropriately dubbed neutrophil extracellular traps (NETs)—also serve the dark side. In their ceaseless struggle to gain advantage, cancer cells in the vicinity of neutrophils are able to emit signals that cause the neutrophils to release their NETs, even in the absence of a local infection. In mouse models of triple-negative breast cancer, a very aggressive subtype, Egeblad’s team linked cancer cells’ hijacking of NETs with metastasis. She hypothesizes that malignant cells mobilize the NETs to chew away at scaffolding that supports tissue—in say a lung or bone, where the resulting holes and crevices can be colonized, metastatic outposts fit for occupancy.
With Michael Goldberg of Dana Farber Cancer Institute, Egeblad has figured out a way to hitch a NET-digesting drug called DNase to tiny spheres called nanoparticles. Used to clear NETs in cystic fibrosis, DNase is FDA approved to be delivered through an inhaler. Attaching the drug to nanoparticles increases its stability, and when delivered in a mouse model of triple-negative breast cancer this version reduced and in some cases prevented metastasis to the lung. The team is optimizing the drug, while thinking carefully about which patients might benefit from it and when to administer it in relation to chemotherapy, which depresses the immune system and thus requires any approach to target these ethereal lattices—usually helpful but sometimes harmful—to be confined to metastatic hotspots.
Written by: Peter Tarr, Senior Science Writer | publicaffairs@cshl.edu | 516-367-8455
About

Mikala Egeblad
Adjunct Professor
Ph.D., University of Copenhagen and the Danish Cancer Society, 2000