Cold Spring Harbor, NY — Scientists at Cold Spring Harbor Laboratory (CSHL) have determined one of the ways in which cancers in remission can spring back into action. This knowledge has inspired a new treatment idea designed to prevent cancer recurrence and metastasis.
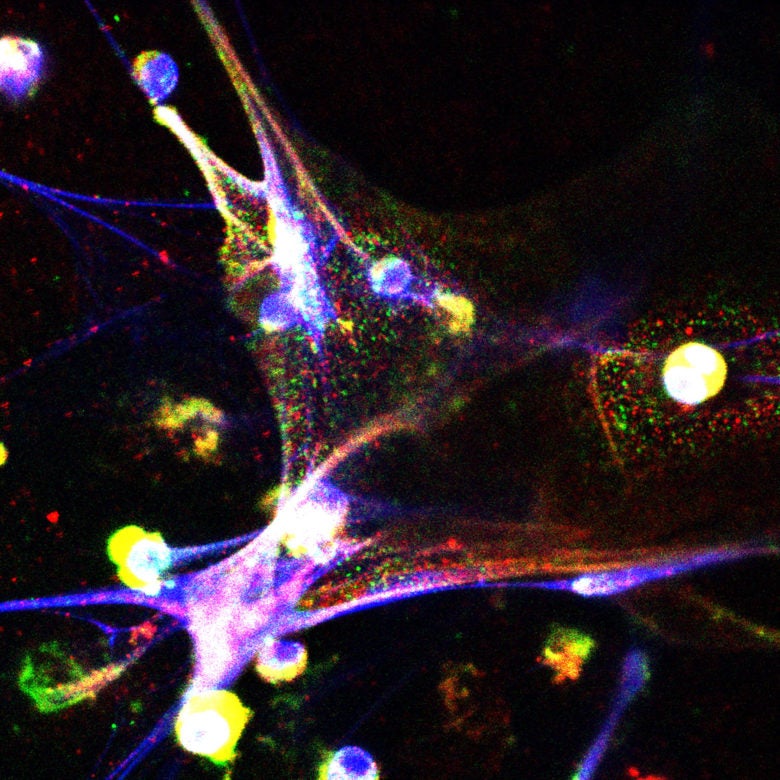
Even after successful cancer treatment, dormant, non-dividing cancer cells that previously detached from the original tumor may still exist elsewhere in the body. If awakened, these cells can proliferate and grow into metastatic tumors. A CSHL team studying metastasis to the lungs has now identified signals accompanying inflammation that can awaken dormant cancer cells.
Whether inflammation can directly cause cancer recurrence, and if so how, has not been clear. In their new research, the team demonstrates that sustained lung inflammation, including that caused by tobacco smoke exposure, can cause dormant breast and prostate cancer cells that have traveled to the lungs to awaken and begin to divide. These cells can now form a metastasis in the lungs. Metastasis accounts for the bulk of lethality from most common cancers.
Importantly, the team, led by CSHL Associate Professor Mikala Egeblad, and including researchers from the Dana-Farber Cancer Institute and UC Davis, also demonstrate a way of blocking the signaling that awakened the dormant cancer cells, a concept that could prevent cancer recurrence or lessen its frequency.
Egeblad’s team showed that sustained lung inflammation, caused either by exposing mice to tobacco smoke or to a component of bacteria known as endotoxin, induced common white blood cells called neutrophils to awaken nearby dormant cancer cells in an extraordinary way.
Neutrophils, which we normally rely upon to kill invaders like bacteria and yeast, have several ways of vanquishing their prey. One is to expel their DNA into the space beyond the cell membrane. Laced with toxic enzymes, this expelled DNA forms gauzy, net-like traps (called neutrophil extracellular traps, or NETs) that can kill a pathogen.
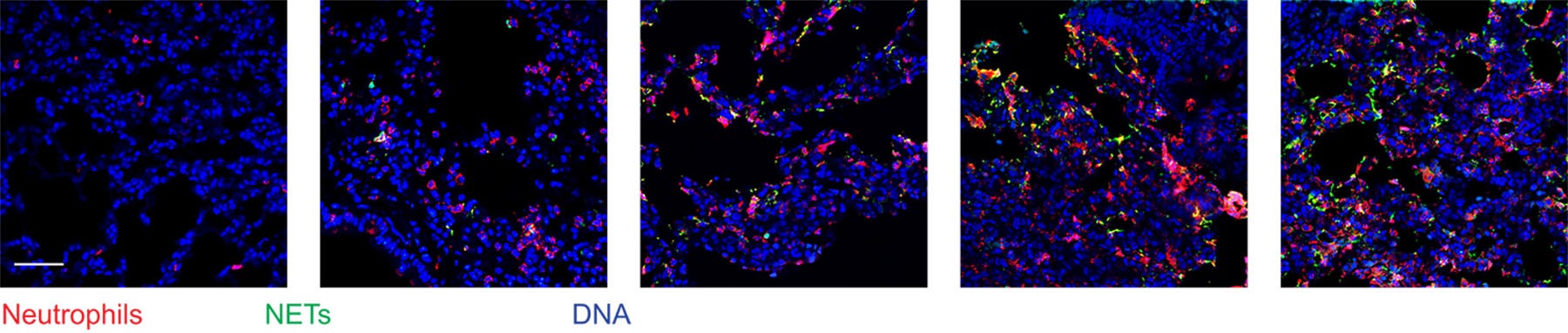
The new research shows that sustained lung inflammation causes the formation of NETs in the area around dormant cancer cells. Two enzymes in the NETs, called NE (neutrophil elastase) and MMP9 (matrix metalloproteinase 9), interact with a protein in tissue called laminin. In sequence, first NE then MMP9 make cuts in laminin proteins. This changes the protein’s shape, exposing a new surface, called an epitope.
This epitope, when recognized by dormant cancer cells nearby, spurs signaling that awakens the cancer cells. “The dormant cancer cells recognize that new shape of the laminin and they say, ‘we should start growing again,’” Egeblad says.
The team created an antibody to block the epitope exposed on the laminin proteins. In mice, this prevented the reawakening of dormant cancer cells nearby. Work has begun to optimize the antibody and compare it with other approaches to interfere with NETs, with the hope of eventually conducting trials in people.
Written by: Peter Tarr, Senior Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
CSHL Cancer Center Support; Department of Defense; Pershing Square Sohn Cancer Research Alliance Prize; Association pour la Recherche sur le Cancer; Terri Brodeur Breast Cancer Foundation (the European Molecular Biology Organization); Susan G. Komen Foundation; Lustgarten Foundation; National Cancer Institute; Cedar Hill Foundation; CSHL and Northwell Health; Starr Centennial Scholarship ; George A. & Marjorie H. Anderson Scholarship; Boehringer Ingelheim Fonds; Deutsche Forschungs Gemeinschaft research fellowship; UC Davis NIEHS Environmental Health Science Center; AACR-Bayer Healthcare Basic Cancer Research Fellowship.
Citation
Albrengues J et al, “Neutrophil extracellular traps produced during inflammation awaken dormant cancer cells in mice” appears online in Science, September 28, 2018.
Principal Investigator

Mikala Egeblad
Adjunct Professor
Ph.D., University of Copenhagen and the Danish Cancer Society, 2000


One thought on “How a sleeping cancer awakens and metastasizes”
Comments are closed.