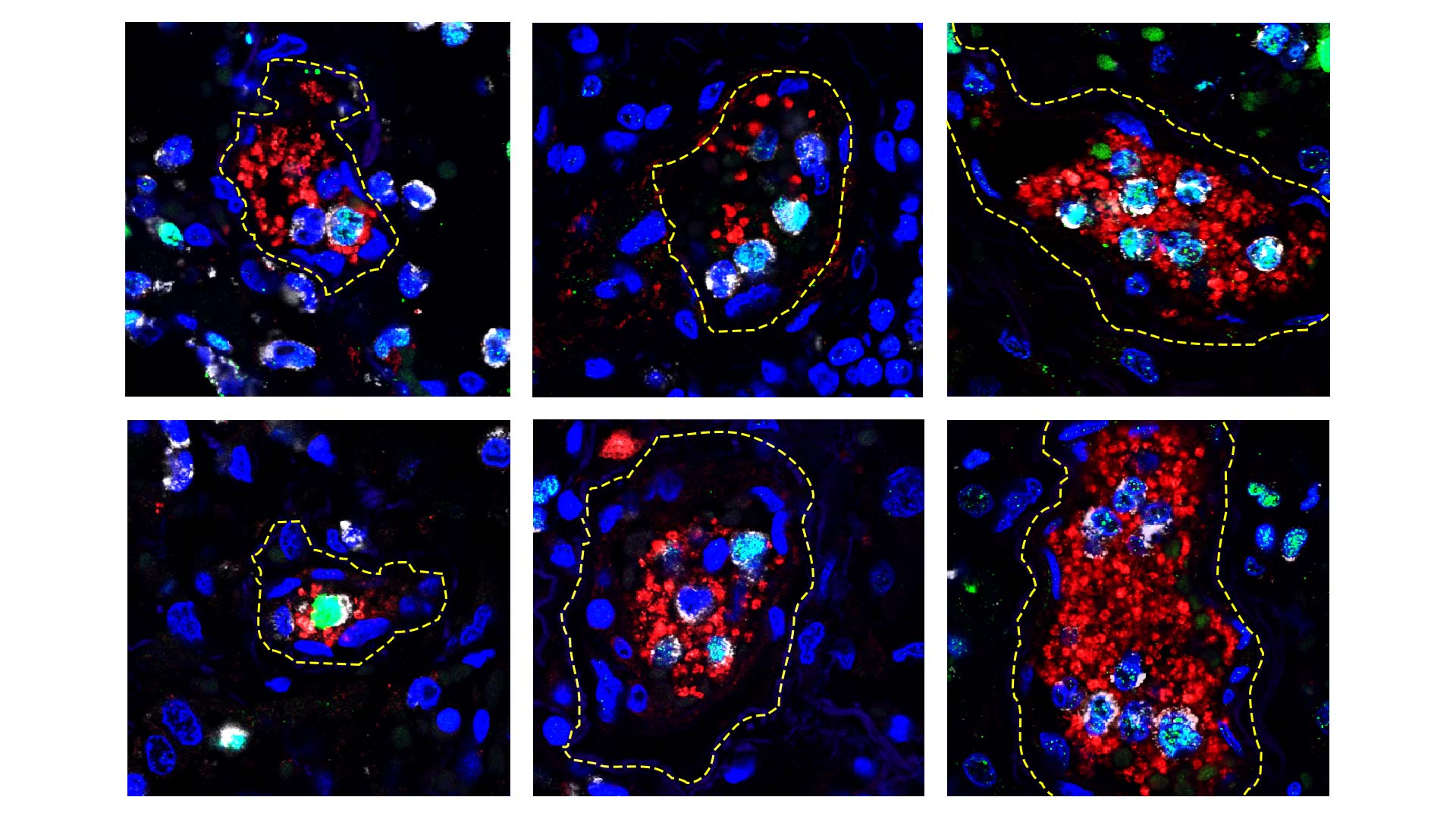
Scientists have new evidence that overactive neutrophils—a common type of circulating immune cell—may drive the life-threatening blood clots and inflammation that occur in some patients with COVID-19. High levels of the sticky, pathogen-trapping webs produced by the cells were associated with the most severe cases of COVID-19 in a study reported online in the journal Blood.
Neutrophil extracellular traps (NETs) are a type of defense that the immune system deploys against certain pathogens—webs of DNA and toxins that ensnare and destroy viruses and bacteria. When too many of these NETs accumulate during a persistent infection, they can lead to acute respiratory distress syndrome, which leads many patients with COVID-19 to require intensive care.
Researchers at Cold Spring Harbor Laboratory (CSHL), University of Utah Health, PEEL Therapeutics and Weill Cornell Medicine collaborated to investigate NETs suspected role in COVID-19, collecting blood samples from 33 hospitalized patients, as well as autopsy tissue. They found that biomarkers of NET formation were more abundant in patients who required ventilation, and highest in the three study participants who eventually died from COVID-19.
When the team examined the lungs of the patients who died, they found tiny clots of tangled NETs and blood platelets known as microthrombi scattered through the tissue. “It will be important to investigate NETs role in clot formation (thrombosis) not only in light of the COVID-19 pandemic, but also to understand their broader role in disease,” says CSHL Associate Professor Mikala Egeblad.
“Excess NETs are formed in other viral diseases,” she says. “We also know that clotting is a major cause of death in people with end-stage cancer, so what we are learning in COVID-19 may help us understand basic properties in cancer and other diseases.”
In the laboratory, neutrophils from patients with COVID-19 churned out exceptionally high levels of NETs, and the researchers found healthy neutrophils behaved the same way when they were exposed to plasma from patients with the illness. They could stop NET production, however, by exposing cells to neonatal NET-Inhibitory Factor (nNIF), an anti-inflammatory peptide from umbilical cord blood that protects newborn babies.
Several therapeutic strategies for dismantling NETs or preventing their formation are currently under investigation. These include the nNIF peptide, which is in pre-clinical development by PEEL Therapeutics. “Although further studies will be required, the NET-inhibitory protein may block exaggerated NET formation in COVID-19 patients,” says Christian Con Yost, whose laboratory at University of Utah Health discovered nNIF in 2016.
Written by: Jennifer Michalowski, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
National Institutes of Health, O’Neill Charitable Trust, Linartz-Meier Family Foundation, University of Utah Triple I Program, Fonds voor Wetenschappelijk Onderzoek Vlaanderen, Animal Cancer Foundation, Soccer For Hope Foundation, Closer To Cure Foundation, PEEL Therapeutics, Inc., and the U.S. Department of Veterans Affairs Clinical Sciences R&D (CSRD) Service.
Citation
Middleton, E. et. al., “Neutrophil Extracellular Traps (NETs) Contribute to Immunothrombosis in COVID-19 Acute Respiratory Distress Syndrome,” Blood, June 29, 2020. DOI: https://doi.org/10.1182/blood.2020007008
About University of Utah Health
University of Utah Health provides leading-edge and compassionate medicine through a system that includes 12 community clinics and four hospitals. A hub for health sciences research and education in the region, U of U Health has a robust research enterprise and trains the majority of Utah’s physicians and health care providers. For 10 straight years, U of U Health has ranked among the top 10 U.S. academic medical centers in the Vizient Quality and Accountability Study.
Contact: Julie Kiefer, julie.kiefer@hsc.utah.edu, 801-597-4258
About PEEL Therapeutics, Inc.
PEEL Therapeutics, Inc. is an emerging biotech that delivers evolution-inspired medicines to improve patient lives. The PEEL pipeline originates from assets inspired by the evolution of elephants, plants and humans for the treatment of cancer and inflammatory diseases, including COVID-19.
Contact: Joshua Schiffman, MD, jschiffman@peeltx.com, 1-650-464-0747
Principal Investigator

Mikala Egeblad
Adjunct Professor
Ph.D., University of Copenhagen and the Danish Cancer Society, 2000