Cold Spring Harbor, NY — Most people have never tasted a groundcherry; the small, sweet relative of the tomato that plant biologist Zachary Lippman describes as “tropically intoxicating.” That’s because the groundcherry plant, with its long, straggly branches and sporadically ripening fruit, is unsuitable for large-scale agriculture. For now, the treat is reserved for people willing to wrangle the plant in their garden or those lucky enough to find it at a farmers’ market.
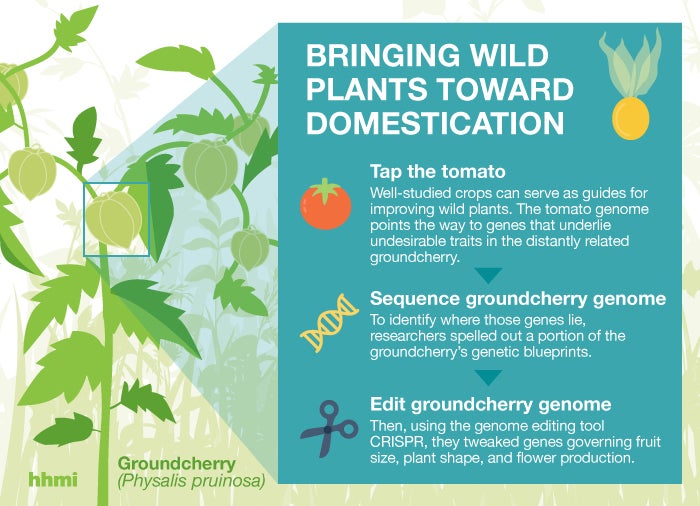
That could change, thanks to the genome-editing tool known as CRISPR. Howard Hughes Medical Institute Investigator and Cold Spring Harbor Laboratory (CSHL) Professor Lippman, heads a team with deep knowledge of tomato plants and the genes that control their growth. They used CRISPR to engineer groundcherries that maintain compact, manageable stems and that produce larger, more abundant fruit.
Lippman says the fruit, which develops inside a papery husk like a tomatillo, has a surprising flavor, echoing both tomato and pineapple. The plant that produces it is still wild: no effort has been made to make it a practical crop through traditional breeding practices.
Because the plant is closely related to the well-studied tomato, Lippman’s team had clues about which genes control the groundcherry’s growth and flowering. “We knew of some clear targets to improve the plant’s architecture, fruit production, and fruit size,” he says.
In the October 1, 2018 issue of Nature Plants, Lippman, collaborator Joyce Van Eck at the Boyce Thompson Institute, and colleagues report the effects of CRISPR-mediated changes to three groundcherry genes. One change reins in production of a hormone that regulates flowering; making plants more compact and producing fruit in clusters rather than one by one. A change to another flowering hormone leads to denser fruit production: plants with a CRISPR-generated mutation in this gene produced up to 50 percent more fruit along a given stem length than the unmodified plant, Lippman says. The third change boosts the number of seedy sections within each fruit, thereby increasing its overall size.
Lippman’s team is working on other aspects of the plant to make the groundcherry practical for large-scale production. The current success demonstrates that it’s possible. In less than two years, they achieved improvements that would have taken far longer using traditional breeding practices.
Lippman is enthusiastic about making groundcherries more widely available. But the implications are much broader. “This is pretty good proof that with gene editing you can think about bringing other wild plants or orphan crops into agricultural production,” he says. “The more arrows we have in our quiver to address agricultural needs in the future, the better off we’re going to be.”
Written by: Jennifer Michalowski, Science Writer | publicaffairs@cshl.edu | 516-367-8455
Funding
NSF Postdoctoral Research Fellowship in Biology; NSF Plant Genome Research Program
Citation
Z. Lemmon et al. ‘Rapid improvement of domestication traits in an orphan crop by genome editing’ was published on October 1, 2018, Nature Plants. DOI: http://dx.doi.org/10.1038/s41477-018-0259-x
Principal Investigator

Zachary Lippman
Professor & HHMI Investigator
Jacob Goldfield Professor of Genetics
Director of Graduate Studies
Ph.D., Watson School of Biological Sciences at Cold Spring Harbor Laboratory, 2004

One thought on “CRISPR could bring groundcherries to market”
Comments are closed.